Nasopharyngeal Carcinoma
Nasopharyngeal carcinoma is a malignant epithelial neoplasm arising from the nasopharyngeal mucosa, strongly associated with Epstein-Barr virus infection, and characterized by early lymph node metastasis and high radiosensitivity.
Nasopharyngeal Carcinoma (NPC)
Nasopharyngeal carcinoma (NPC) is a malignant epithelial neoplasm arising from the mucosal lining of the nasopharynx — the uppermost part of the pharynx, situated directly behind the nasal cavity and above the soft palate. The term breaks down as: "naso" = nose, "pharyngeal" = relating to the pharynx (throat), "carcinoma" = epithelial-origin cancer.
NPC is biologically and epidemiologically distinct from other head and neck squamous cell carcinomas (HNSCCs). Unlike most HNSCCs that are driven primarily by tobacco/alcohol, NPC is uniquely associated with Epstein-Barr virus (EBV) infection [1][2], has a striking geographic predilection for Southern China (including Hong Kong), and is exquisitely radiosensitive — meaning radiation therapy (RT), not surgery, is the primary treatment modality.
Why is NPC different from other H&N cancers?
NPC stands apart because of: (1) its unique viral etiology (EBV, not HPV), (2) its geographic/ethnic clustering, (3) the nasopharynx's deep anatomical location making surgical access extremely difficult, (4) its remarkable radiosensitivity, and (5) its distinct N staging system (different from all other H&N cancers) [3].
2. Epidemiology
- > 70% of new NPC cases occur in East and Southeast Asia [1]
- China alone accounts for ~47.7% of global cases (60,558 out of ~127,000 cases globally per GLOBOCAN 2018 data) [1]
- Other high-burden countries: Indonesia (14.2%), Vietnam (4.9%), India (4.0%), Mediterranean countries (2.9%), Philippines (2.3%), Thailand (1.7%), USA (1.7%) [1]
- Endemic regions form a characteristic belt: Southern China > Southeast Asia > North Africa/Mediterranean > Arctic (Inuit populations)
- Global gradual decline in incidence — attributed to changes in dietary habits, improved socioeconomic conditions, and reduced consumption of preserved foods [1]
- NPC is the 10th most common cancer in Hong Kong (both sexes combined) [1][2]
- 6th most common cancer in males in Hong Kong [2]
- Male-to-female ratio = 2.5:1 [1] (some sources cite 2–3:1 [2])
- Peak incidence: bimodal — small peak at age 15–25 years, major peak at age 40–60 years
- Cantonese-speaking populations in Guangdong province and Hong Kong have the highest incidence worldwide (age-standardised rate ~20–30 per 100,000 in males)
- Even when Southern Chinese emigrate to lower-risk areas, first-generation migrants retain elevated risk, which gradually declines in subsequent generations — supporting both genetic susceptibility and environmental/dietary factors
High Yield: NPC is endemic in Southern China. In Hong Kong, it is the 10th most common cancer overall and 6th in males. The male predominance is approximately 2.5:1. Always think NPC when a young-to-middle-aged Cantonese male presents with a neck mass or nasal symptoms.
3. Anatomy and Function of the Nasopharynx
Understanding the anatomy is absolutely critical because the pattern of local invasion, cranial nerve involvement, and lymphatic drainage dictates the clinical presentation.
| Boundary | Structure |
|---|---|
| Superior (roof) | Body of sphenoid bone, basilar part of occipital bone (clivus) |
| Posterior wall | C1–C2 vertebral bodies, prevertebral fascia |
| Lateral walls | Pharyngeal recesses (fossae of Rosenmüller), Eustachian tube openings |
| Inferior | Superior surface of soft palate (nasopharynx communicates with oropharynx at the level of the soft palate) |
| Anterior | Posterior choanae (openings into nasal cavity) |
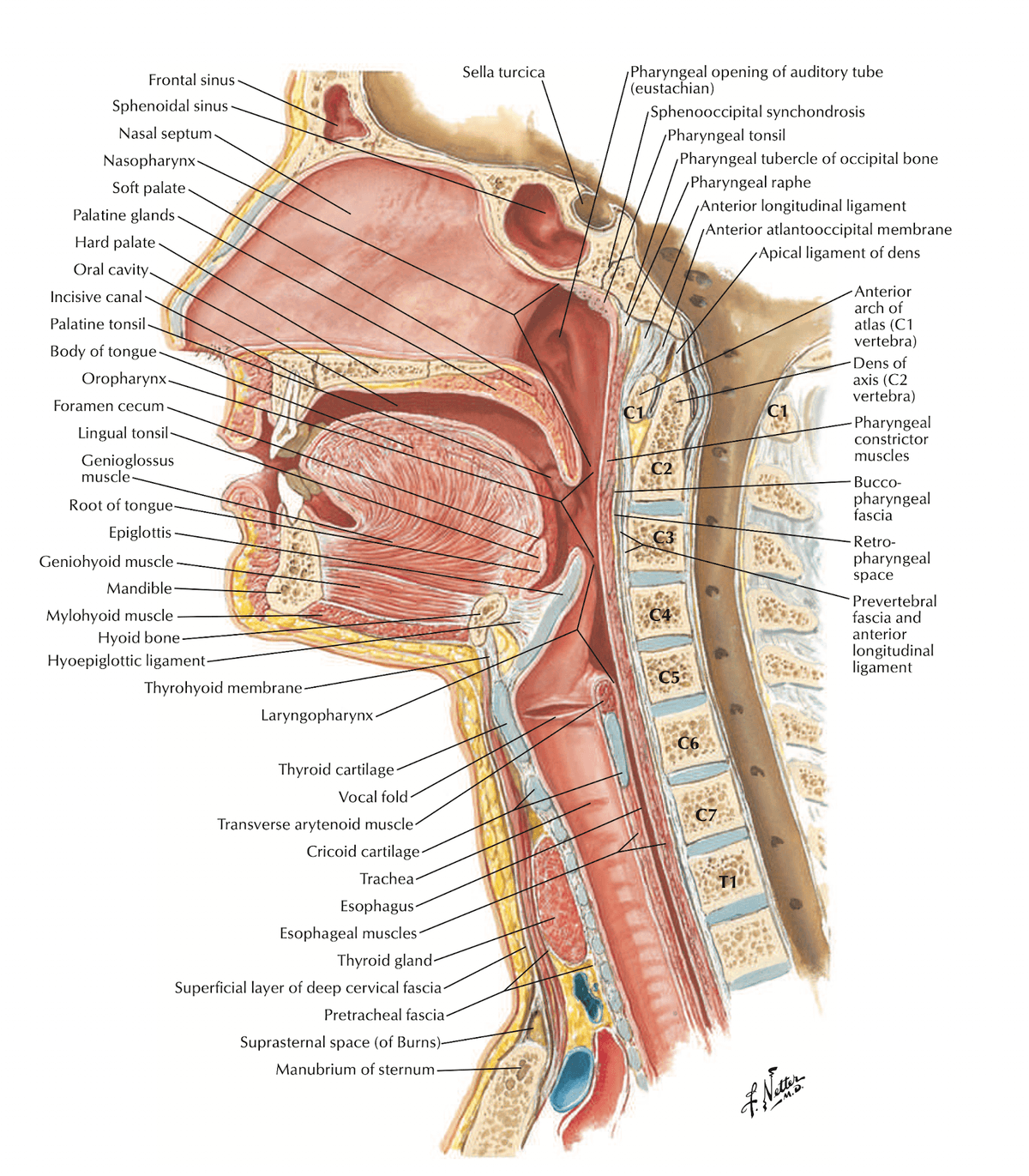
-
Pharyngeal recess (Fossa of Rosenmüller): A lateral mucosal recess just behind the torus tubarius (the cartilaginous elevation around the Eustachian tube opening). This is the most common site of origin of NPC [2]. Why here? The fossa has abundant lymphoid tissue and is a transitional zone between respiratory and squamous epithelium — transition zones are generally vulnerable to malignant transformation (similar concept to the squamocolumnar junction in cervical cancer).
-
Eustachian tube opening (torus tubarius): Located on the lateral wall. Tumour infiltration here causes Eustachian tube dysfunction → middle ear effusion → conductive hearing loss. This is one of the earliest symptoms.
-
Levator veli palatini and tensor veli palatini: Muscles that open the Eustachian tube. Tumour invasion of these muscles also contributes to tubal dysfunction.
The nasopharynx sits in a "crossroads" position at the skull base, surrounded by vital neurovascular structures:
Routes of Local Spread
| Route | Structures Involved | Clinical Consequence |
|---|---|---|
| Lateral → Parapharyngeal space | Fat, internal carotid artery (ICA), CN IX–XII | Cranial nerve palsies (IX–XII), carotid encasement |
| Superolateral → Foramen ovale | CN V3 (mandibular branch of trigeminal) | Facial numbness (V3 distribution), masticator muscle weakness |
| Superior → Foramen lacerum → Cavernous sinus | CN III, IV, V1, V2, VI; ICA; pituitary | Diplopia, ptosis, facial numbness (V1/V2), ophthalmoplegia |
| Superior → Clivus / Sphenoid sinus | Bone erosion | Headache, CSF leak (rare) |
| Anterior → Nasal cavity, pterygopalatine fossa, orbit | Nasal obstruction, orbital extension | Epistaxis, nasal obstruction, proptosis |
| Posterior → Prevertebral muscles, cervical spine | Retropharyngeal nodes, vertebral destruction | Neck stiffness, retropharyngeal mass |
The nasopharynx has an extremely rich lymphatic network, which is why lymph node metastasis is present at diagnosis in up to 75–90% of patients [2]:
- Retropharyngeal lymph nodes (nodes of Rouvière): First echelon drainage — these are the most commonly involved nodes and are clinically occult (cannot be palpated; only detected on imaging)
- Level II (upper deep cervical / jugulodigastric): Most common clinically palpable nodal station
- Level III, IV, V: Progressive drainage
- Bilateral nodal involvement is common because the nasopharynx is a midline structure with bilateral lymphatic drainage
Clinical Pearl – Why does NPC present late?
The nasopharynx is a clinically occult site [2] — you cannot see it without a mirror or endoscope. The fossa of Rosenmüller is hidden behind the torus tubarius. Additionally, the earliest lymph nodes involved (retropharyngeal) are deep and impalpable. By the time the patient notices a neck lump (level II node) or develops cranial nerve symptoms (skull base invasion), the disease is already locally or regionally advanced.
4. Etiology and Risk Factors
NPC has a multifactorial etiology involving the classic triad: viral (EBV) + genetic susceptibility + environmental factors.
EBV infection is the primary etiological agent in the pathogenesis of NPC [1][2]
-
EBV (Human Herpesvirus 4) is a ubiquitous gamma-herpesvirus that infects > 90% of the world's adult population. Most people harbour latent EBV asymptomatically. So why does NPC develop in only a tiny fraction?
- The answer lies in the interplay between EBV latent gene expression, host genetic susceptibility, and environmental co-carcinogens
-
Mechanism of EBV-driven carcinogenesis:
- EBV infects nasopharyngeal epithelial cells (likely via cell-to-cell contact with EBV-infected B lymphocytes in the subepithelial lymphoid tissue)
- The virus establishes latent infection (Latency Type II pattern) expressing:
- LMP1 (Latent Membrane Protein 1): Mimics constitutively active CD40 receptor → activates NF-κB, MAPK, PI3K/Akt pathways → promotes cell survival, proliferation, and anti-apoptosis
- LMP2A: Mimics B-cell receptor signalling → promotes survival
- EBNA1 (EBV Nuclear Antigen 1): Required for episomal maintenance and replication of EBV genome
- EBERs (EBV-encoded small RNAs): Most abundantly expressed; used for diagnosis via in situ hybridization (EBER-ISH) — the gold standard for confirming EBV in tumour tissue
- BARTs (BamHI-A Rightward Transcripts): Encode miRNAs that promote immune evasion
- These viral oncoproteins cooperate with pre-existing genetic/epigenetic alterations (e.g., loss of p16/CDKN2A, chromosome 3p deletions) to drive malignant transformation
-
Serological evidence [2]:
- IgA antibodies against EBV viral capsid antigen (EBV VCA-IgA) — elevated in NPC patients; used as a serological screening marker
- IgA against EBV Early Antigen (EA-IgA) — also elevated
- These are IgA (not IgG) because the nasopharynx is a mucosal surface → local mucosal immune response produces IgA
-
Detection of EBV DNA and EBV gene expression in precursor lesions and tumour cells [2] — confirms that EBV is not a bystander but causally involved
-
Plasma EBV DNA (cell-free EBV DNA): A critically important biomarker:
- Used for screening (Hong Kong population screening studies by Prof Dennis Lo's group)
- Used for staging (levels correlate with tumour burden)
- Used for monitoring treatment response and detecting relapse
- Derived from tumour cell apoptosis/necrosis releasing EBV DNA fragments into blood
High Yield: EBV is detected in virtually 100% of non-keratinizing NPC in endemic areas. The key serological markers are VCA-IgA and EA-IgA. Plasma EBV DNA is now used for population screening, prognostication, and surveillance.
Family history of NPC is a significant risk factor [1][2]:
- First-degree relatives of NPC patients have a 4–10x increased risk
- Associated with certain human leukocyte antigen (HLA) haplotypes [2]:
- HLA-A2, HLA-B46, HLA-B58 — associated with increased risk in Southern Chinese
- HLA-A11 — may be protective
- HLA molecules determine which EBV peptides are presented to cytotoxic T-cells; certain haplotypes may impair immune surveillance against EBV-infected cells
- Associated with genetic polymorphisms such as CYP2A6 [2]:
- CYP2A6 is a polymorphism of the nitrosamine-metabolizing gene — certain variants may reduce the ability to detoxify dietary nitrosamines, increasing mucosal exposure to carcinogens
- GWAS studies have identified susceptibility loci on chromosomes 6p21 (HLA region), 3p21, 9p21 (CDKN2A/p16 locus)
4.3 Environmental Factors
- Salted fish (Cantonese-style salted fish, in particular) [2]
- Classified as a Group 1 carcinogen by IARC
- Contains high levels of nitrosamines, bacterial mutagens, direct genotoxins, and EBV-reactivating substances [2]
- Why salted fish specifically? The traditional Cantonese salt-curing process (partial drying at moderate temperature) promotes bacterial fermentation → generates volatile N-nitrosamines (e.g., N-nitrosodimethylamine, NDMA)
- Cooking of salt-cured food releases volatile nitrosamines which are carried by steam and distributed over the nasopharyngeal mucosa [2]
- Early childhood exposure appears particularly important (developing nasopharyngeal mucosa may be more susceptible)
- Preserved or fermented foods [1][2] — preserved vegetables, fermented bean paste, etc.
- Active and passive tobacco smoking [1] is a risk factor
- Smoking is the primary risk factor for most H&N cancers [4], but for NPC specifically, it acts as a co-carcinogen alongside EBV
- Tobacco smoke contains nitrosamines and polycyclic aromatic hydrocarbons that can directly damage nasopharyngeal mucosa and reactivate latent EBV
- Synergism between smoking and alcohol in HNSCC development is well established [4]
- Alcohol [1] — a classical risk factor for H&N cancer
- Ethanol is metabolized to acetaldehyde (a Group 1 carcinogen) by alcohol dehydrogenase
- East Asian populations frequently carry the ALDH2*2 polymorphism ("Asian flush") → impaired acetaldehyde metabolism → prolonged exposure to this carcinogen
- Oral hygiene [1] — poor oral hygiene with chronic mucosal inflammation may act as a co-carcinogen
- Occupational exposures: wood dust, formaldehyde (less well-established for NPC compared to sinonasal cancers)
- Previous irradiation [4] — prior radiotherapy to H&N region increases risk of subsequent malignancy
Risk Factors Mnemonic for NPC
Think "EBV-GPS":
- E = EBV infection (primary agent)
- B = Brine (salted fish, preserved foods)
- V = Vulnerability genes (HLA haplotypes, CYP2A6, family history)
- G = Geography (Southern China, Cantonese)
- P = Passive/active smoking
- S = Spirits (alcohol)
5. Pathophysiology
NPC does not arise de novo. It follows a stepwise progression, with EBV infection being a relatively late but essential event:
Key molecular events:
- Early events (pre-EBV): Loss of chromosome 3p (contains tumour suppressor genes), inactivation of p16/CDKN2A (cell cycle regulator) by promoter hypermethylation
- EBV infection: EBV preferentially infects pre-malignant epithelium that has already lost key tumour suppressor functions
- Late events (post-EBV): LMP1-driven NF-κB activation, telomerase activation, further genomic instability, immune evasion via PD-L1 upregulation
This is a commonly asked conceptual question. Several reasons:
- The nasopharynx is rich in lymphoid tissue (pharyngeal tonsil/adenoid) → abundant EBV-infected B lymphocytes in close proximity to epithelial cells → facilitates cell-to-cell EBV transmission to epithelial cells
- The fossa of Rosenmüller represents a transitional epithelial zone (respiratory → squamous) — transition zones are inherently more vulnerable to neoplastic transformation
- Direct exposure to inhaled/ingested carcinogens (nitrosamines from salted fish cooking steam) contacts the nasopharyngeal mucosa first
NPC (particularly the undifferentiated subtype) is characterized by a dense lymphoid infiltrate — historically called "lymphoepithelioma" because the tumour cells are intimately admixed with non-neoplastic lymphocytes. This is important because:
- The lymphocytic infiltrate represents an anti-tumour immune response (mostly CD8+ T cells)
- NPC tumour cells evade this response by upregulating PD-L1 (programmed death-ligand 1) → binds PD-1 on T cells → immune checkpoint inhibition → T cell exhaustion
- This is the rationale for immune checkpoint inhibitors (anti-PD-1: pembrolizumab, nivolumab, camrelizumab) in the treatment of advanced/recurrent NPC
6. Classification
The WHO classification divides NPC into three types [1][2]:
| WHO Type | Description | Key Features |
|---|---|---|
| Non-keratinizing | Undifferentiated | Most common endemic form of NPC (Hong Kong) [1][2]. Poorly differentiated, syncytial growth pattern. Strongly associated with EBV infection [1][2]. Dense lymphocytic infiltrate ("lymphoepithelioma"). Possesses a more favourable prognosis than other types [2] — highly radiosensitive |
| Non-keratinizing | Differentiated | Shows evidence of squamous maturation but no overt keratin. Also EBV-associated. Intermediate behaviour |
| Keratinizing squamous cell carcinoma | Most common sporadic form of NPC [2]. Resembles cell of origin (squamous cell carcinoma) [2]. Produces keratin. More common in non-endemic areas. Less strongly associated with EBV. Worse prognosis than non-keratinizing | |
| Basaloid squamous cell carcinoma | Rare. Aggressive clinical course. Poor survival and prognosis [2] |
- > 95% of cases in endemic areas are non-keratinizing [1] (vast majority undifferentiated)
- Epstein-Barr virus infection is found in virtually all non-keratinizing NPC in endemic areas [1]
Exam Pearl – WHO Classification
Students often confuse the old WHO classification (Type I/II/III) with the current system. The old Type III ("undifferentiated") is now called non-keratinizing undifferentiated. The current WHO classification (2017) dropped the numeric system. Use the descriptive terms: non-keratinizing (differentiated/undifferentiated), keratinizing SCC, and basaloid SCC.
6.2 TNM Staging (AJCC/UICC 8th Edition, 2017)
TNM staging for NPC is distinct from other H&N cancers [3], particularly the N staging.
| T Stage | Description |
|---|---|
| Tis | Carcinoma in situ |
| T1 | Tumour confined to nasopharynx, OR extends to oropharynx and/or nasal cavity WITHOUT parapharyngeal involvement |
| T2 | Tumour with extension to parapharyngeal space, AND/OR adjacent soft tissue involvement (medial pterygoid, lateral pterygoid, prevertebral muscles) |
| T3 | Tumour involves bony structures of skull base, cervical vertebrae, pterygoid structures, AND/OR paranasal sinuses |
| T4 | Tumour with intracranial extension, involvement of cranial nerves, hypopharynx, orbit, parotid gland, AND/OR extensive soft tissue infiltration beyond the lateral surface of the lateral pterygoid muscle |
N staging for NPC is different from all other H&N cancers [3]. It does NOT use the ENE (extranodal extension) criteria used for other H&N sites. Instead, it uses size and laterality as well as anatomic location:
| N Stage | Description |
|---|---|
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Unilateral cervical and/or uni/bilateral retropharyngeal lymph node(s), ≤ 6 cm above the caudal border of the cricoid cartilage |
| N2 | Bilateral cervical lymph node(s), ≤ 6 cm above the caudal border of the cricoid cartilage |
| N3 | Lymph node(s) > 6 cm AND/OR extension below the caudal border of the cricoid cartilage |
Important: For NPC, N staging is based on (1) unilateral vs bilateral, (2) size (≤ 6 cm vs > 6 cm), and (3) position relative to the caudal border of the cricoid cartilage (supraclavicular involvement). This is completely different from the uniform N staging used for other H&N cancers [3].
| M Stage | Description |
|---|---|
| M0 | No distant metastasis |
| M1 | Distant metastasis present |
| Stage | T | N | M |
|---|---|---|---|
| I | T1 | N0 | M0 |
| II | T1 N1, T2 N0-1 | M0 | |
| III | T1-2 N2, T3 N0-2 | M0 | |
| IVA | T4 or N3 | M0 | |
| IVB | Any T | Any N | M1 |
Common sites of distant metastasis [2]:
- Bone (most common, ~75%) — Why? NPC has a tropism for bone marrow, possibly related to CXCR4/CXCL12 chemokine axis
- Liver
- Lung
- Distant lymph nodes
- Less commonly: brain, skin, adrenal
7. Clinical Features
NPC remains asymptomatic for a long period due to its presence in a clinically occult site [2]. The patient typically presents with locally or regionally advanced disease due to the prolonged asymptomatic period or due to missed diagnosis [2].
The clinical features can be organized by the direction of tumour spread:
7.2 Symptoms
| Symptom | Pathophysiological Basis |
|---|---|
| Epistaxis (blood-stained nasal discharge / post-nasal drip) | Tumour neovasculature is friable → bleeds easily. Tumour extends anteriorly into the nasal cavity or erodes mucosal blood vessels. Often blood-stained post-nasal drip rather than frank anterior epistaxis |
| Nasal obstruction (unilateral initially, may become bilateral) | Physical mass effect of tumour obstructing the posterior choana. May also extend anteriorly into the nasal cavity |
| Rhinorrhoea | Mucosal irritation and secondary infection/inflammation around the tumour |
| Symptom | Pathophysiological Basis |
|---|---|
| Unilateral serous otitis media (middle ear effusion) | Tumour infiltrates or compresses the Eustachian tube opening (torus tubarius) or its associated muscles (levator/tensor veli palatini) → loss of Eustachian tube function → failure to equalize middle ear pressure → transudation of fluid into middle ear. This is one of the earliest symptoms and is a critical diagnostic clue |
| Conductive hearing loss (unilateral) | Middle ear effusion dampens tympanic membrane vibration and ossicular chain transmission. Any adult with unexplained unilateral middle ear effusion must have the nasopharynx examined to exclude NPC |
| Tinnitus (usually low-pitched) | Fluid in the middle ear alters resonance characteristics |
| Ear fullness / aural blockage | Negative middle ear pressure from Eustachian tube dysfunction |
Critical Rule
Any adult in an NPC-endemic area presenting with unilateral serous otitis media (middle ear effusion) MUST have the nasopharynx examined (by nasopharyngoscopy) to exclude NPC. In children, serous otitis media is common and usually due to adenoid hypertrophy. In adults, it is uncommon and NPC must be ruled out.
Cranial nerve (CN) involvement occurs via two main routes:
- Through the foramen lacerum → cavernous sinus (CN III, IV, V1, V2, VI)
- Through the parapharyngeal space (CN IX, X, XI, XII)
- Through the foramen ovale (CN V3)
| Symptom | Cranial Nerve | Pathophysiological Basis |
|---|---|---|
| Diplopia | CN VI (abducens) — most commonly affected CN in NPC | CN VI has the longest intracranial course and passes through the cavernous sinus → vulnerable to compression. Lateral rectus palsy → inability to abduct the ipsilateral eye → horizontal diplopia |
| Diplopia with ptosis | CN III (oculomotor) | Passes through the cavernous sinus. Innervates superior/inferior/medial rectus, inferior oblique, levator palpebrae superioris, and parasympathetics to pupil. Compression → ptosis + "down and out" eye + mydriasis |
| Facial numbness / pain | CN V (trigeminal) — V2 and V3 most commonly | V2 passes through foramen rotundum into the cavernous sinus → cheek/upper lip numbness. V3 passes through foramen ovale → lower face/jaw numbness. Perineural spread along V is also possible |
| Headache | CN V (V1/V2) + Dural invasion | Dural stretch/invasion at the skull base → referred pain. V1 innervates the tentorium; frontal headache suggests V1 involvement |
| Trismus (difficulty opening mouth) | CN V3 motor branch / Direct invasion of pterygoid muscles | Motor V3 innervates muscles of mastication (masseter, temporalis, medial/lateral pterygoids). Invasion or compression → weakness. Direct tumour invasion of pterygoid muscles also causes mechanical restriction |
| Hoarseness | CN X (vagus) | Recurrent laryngeal nerve branch of vagus innervates all intrinsic laryngeal muscles except cricothyroid. Vagal palsy → vocal cord paralysis → breathy hoarse voice |
| Dysphagia | CN IX (glossopharyngeal), CN X | Pharyngeal motor innervation disrupted → pharyngeal weakness → difficulty swallowing |
| Shoulder weakness / trapezius wasting | CN XI (accessory) | Innervates sternocleidomastoid and trapezius. Parapharyngeal space invasion → shoulder drop |
| Tongue deviation | CN XII (hypoglossal) | Innervates all intrinsic and extrinsic tongue muscles except palatoglossus. Unilateral palsy → tongue deviates toward the affected side (the functional side pushes the tongue over) |
Teaching Pearl: The most commonly affected cranial nerve in NPC is CN V (trigeminal) followed by CN VI (abducens). When CN III, IV, V1, V2, and VI are involved together, this suggests cavernous sinus syndrome. When CN IX, X, XI, XII are involved, this points to parapharyngeal space / jugular foramen syndrome (Vernet syndrome).
| Symptom | Pathophysiological Basis |
|---|---|
| Painless neck mass — often the presenting complaint | Metastasis to cervical lymph nodes, most commonly level II (upper jugular / jugulodigastric). Lymph node metastases are usually present at diagnosis and commonly bilateral [2] because of the midline position of the nasopharynx with bilateral lymphatic drainage. The mass is typically firm, non-tender, and may be fixed to underlying structures in advanced cases |
Presenting Complaint Statistics
In endemic areas, the most common presenting symptom of NPC is a neck mass (up to 40-70% of patients), followed by nasal symptoms (epistaxis, obstruction) and otological symptoms (hearing loss, ear fullness). Cranial nerve symptoms indicate advanced local disease (T3-T4).
| Symptom | Pathophysiological Basis |
|---|---|
| Bone pain (back, pelvis, ribs) | Bone is the most common distant metastatic site. Tumour cells in bone marrow stimulate osteoclast activity → bone destruction → pathological fractures → pain |
| Hepatomegaly / right upper quadrant pain | Liver metastasis → capsular stretch |
| Cough / haemoptysis | Lung metastasis |
| Constitutional symptoms (weight loss, fatigue, anorexia) | Tumour-related cachexia mediated by cytokines (TNF-α, IL-6) |
7.3 Signs
| Sign | Basis |
|---|---|
| Nasopharyngeal mass on posterior rhinoscopy / nasopharyngoscopy | Direct visualization of the primary tumour, most commonly at the fossa of Rosenmüller. May appear as an exophytic mass, mucosal irregularity, or submucosal bulge |
| Blood-stained mucus in post-nasal space | Tumour surface ulceration/friable vasculature |
| Dull tympanic membrane / middle ear effusion (on otoscopy) | Eustachian tube obstruction → serous otitis media. Tympanometry shows Type B (flat) curve |
| Conductive hearing loss (on tuning fork / audiometry) | Weber lateralizes to the affected ear; Rinne negative on affected side |
| Sign | Basis |
|---|---|
| Cervical lymphadenopathy (firm, non-tender, may be bilateral) | Lymph node metastasis are usually present at diagnosis and commonly bilateral [2]. Most commonly at level II (jugulodigastric). Can be massive (> 6 cm). Fixed nodes suggest extracapsular spread |
| Posterior triangle lymphadenopathy (Level V) | Less common but important — NPC is one of the few H&N cancers that commonly metastasizes to level V |
| Sign | CN Involved | Examination Finding |
|---|---|---|
| Lateral rectus palsy | CN VI | Failure of abduction of the ipsilateral eye; convergent strabismus |
| Complete ophthalmoplegia with ptosis | CN III, IV, VI (cavernous sinus syndrome) | Eye "frozen" in position, ptosis, dilated pupil |
| Facial sensory loss | CN V (V1, V2, V3) | Absent corneal reflex (V1), reduced sensation over cheek (V2) or jaw (V3) |
| Horner syndrome | Sympathetic chain in parapharyngeal space | Miosis, ptosis, anhidrosis — indicates parapharyngeal invasion involving the cervical sympathetic chain |
| Palatal deviation | CN X (vagus) | Uvula deviates to the normal side; palate fails to elevate on the affected side |
| Vocal cord paralysis (on laryngoscopy) | CN X | Immobile vocal cord on affected side |
| Tongue deviation and wasting | CN XII | Tongue deviates to the affected side; ipsilateral atrophy and fasciculations |
| Trapezius weakness | CN XI | Shoulder drop, inability to shrug shoulder against resistance |
| Sign | Basis |
|---|---|
| Proptosis | Direct orbital invasion (rare, advanced T4 disease) |
| Visual field defect (bitemporal hemianopia) | Extension to optic chiasm via cavernous sinus (very rare) |
- Dermatomyositis: NPC is one of the malignancies associated with paraneoplastic dermatomyositis, particularly in Chinese populations
- Hypertrophic pulmonary osteoarthropathy: Rare
| Direction of Spread | Structures Involved | Symptoms & Signs |
|---|---|---|
| Anteriorly | Nasal cavity, posterior choanae | Epistaxis, nasal obstruction |
| Laterally | Eustachian tube | Serous otitis media, conductive hearing loss, tinnitus |
| Laterally (deep) | Parapharyngeal space, CN IX-XII, sympathetic chain | Lower CN palsies, Horner syndrome |
| Superiorly | Skull base, cavernous sinus, CN III-VI | Headache, diplopia, facial numbness, ophthalmoplegia |
| Posteriorly | Retropharyngeal nodes, prevertebral muscles | Retropharyngeal mass, neck stiffness |
| Regionally | Cervical lymph nodes (bilateral) | Painless neck mass |
| Distantly | Bone, liver, lung | Bone pain, hepatomegaly, cough, weight loss |
8. Important Associations and Concepts
While this concept is more relevant to smoking/alcohol-driven H&N cancers than to EBV-driven NPC, it is still worth noting:
- Diffuse and chronic exposure of mucosa of the upper aerodigestive tract to carcinogenic substances leads to widespread changes in mucosal epithelium [3]
- Leads to development of separate tumours at different anatomical sites [3]
- Synchronous tumour = second primary tumour detected within 6 months [3]
- Metachronous tumour = second primary tumour detected > 6 months [3]
- Panendoscopy is always recommended [3] — includes direct laryngoscopy, bronchoscopy, and OGD
- "ALWAYS protect the airway for all H&N cancer" [3] — tumour growth, post-treatment oedema, or haemorrhage can compromise the airway
- NPC rarely causes acute airway obstruction (unlike laryngeal or hypopharyngeal cancers), but post-radiation oedema or bulky retropharyngeal disease can contribute
- H&N cancer treatment affects critical functions: swallowing, speech, breathing, and appearance [5]
- This is especially relevant for NPC because radiation to the nasopharynx inevitably irradiates surrounding structures (salivary glands, temporomandibular joints, cranial nerves, brain)
- Long-term sequelae include xerostomia, osteoradionecrosis, trismus, dysphagia, cranial neuropathy, and temporal lobe necrosis
High Yield Summary
Definition: Malignant epithelial neoplasm of the nasopharynx, most commonly arising from the fossa of Rosenmüller.
Epidemiology: Endemic in Southern China/Hong Kong. 10th most common cancer in HK, 6th in males. M:F = 2.5:1. > 70% of global cases in East/Southeast Asia.
Etiology triad: (1) EBV (primary agent; detected in nearly 100% of non-keratinizing NPC; key markers: VCA-IgA, plasma EBV DNA), (2) Genetics (HLA haplotypes, CYP2A6, family history), (3) Environmental (salted fish/preserved foods with nitrosamines, smoking, alcohol).
Pathology: > 95% non-keratinizing in endemic areas (undifferentiated most common — best prognosis, most radiosensitive). Keratinizing = sporadic form. Basaloid = worst prognosis.
Key anatomy: Nasopharynx → fossa of Rosenmüller (commonest origin). Lateral spread → Eustachian tube (effusion), parapharyngeal space (CN IX-XII, Horner). Superior spread → skull base, cavernous sinus (CN III-VI). Rich lymphatic drainage → early bilateral LN metastasis.
Clinical features:
- Neck mass (most common presenting complaint) — bilateral cervical LN, especially level II
- Nasal: epistaxis, obstruction
- Otological: unilateral serous otitis media (critical clue in adults), conductive hearing loss
- Neurological: CN VI palsy (most common CN), CN V numbness, cavernous sinus syndrome
- Distant metastasis: bone (75%) > liver > lung
Red flag: Any adult with unilateral middle ear effusion in an NPC-endemic region → must scope the nasopharynx.
N staging is unique to NPC — based on laterality, size, and position relative to cricoid (not ENE like other H&N cancers).
Active Recall - Nasopharyngeal Carcinoma (Definition to Clinical Features)
[1] Lecture slides: GC 215. Common nasal conditions and nasopharyngeal carcinoma (1).pdf (p47–50) [2] Senior notes: felixlai.md (felix:357–358) — Nasopharyngeal cancer (NPC) [3] Senior notes: felixlai.md (felix:342, 345) — Head and neck cancer overview, TNM staging [4] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p41) — Etiology of pharyngeal/oral cavity cancers [5] Lecture slides: GC 187. Head and neck cancer problems Function and shape.pdf [6] Image credit: Netter's (nasopharynx anatomy image)
Differential Diagnosis of Nasopharyngeal Carcinoma
When a patient presents with the clinical features described in the previous section — a combination of nasal symptoms (epistaxis, obstruction), otological symptoms (unilateral middle ear effusion), cranial nerve palsies, and/or a neck mass — you need to think systematically. The differential diagnosis is organized around two overlapping clinical scenarios: (A) the nasopharyngeal mass itself and (B) the presenting symptom (most commonly a neck mass, since NPC often presents with cervical lymphadenopathy as the first complaint).
The key conceptual framework: NPC can mimic other nasopharyngeal pathology, and conversely, other conditions can mimic NPC. You need to differentiate by tissue of origin, anatomical location, clinical behaviour, and epidemiological context.
A. Differential Diagnosis of a Nasopharyngeal Mass
These are conditions that produce a mass or abnormal tissue in the nasopharynx itself, and therefore must be distinguished from NPC on endoscopy and biopsy.
| Condition | Key Distinguishing Features | Why it mimics NPC |
|---|---|---|
| Adenoid hypertrophy | Common in children and adolescents. Lymphoid tissue hypertrophy at the nasopharyngeal roof. Regresses after puberty. Causes bilateral nasal obstruction, mouth breathing, snoring. | Produces a midline nasopharyngeal mass on endoscopy, but the tissue is smooth, symmetric, and lymphoid — not ulcerated or irregular. In adults, persistent or recurrent adenoidal tissue should raise suspicion for lymphoma or NPC |
| Tornwaldt cyst (pharyngeal bursa cyst) | Benign cyst arising from a remnant of the notochord in the midline posterior nasopharyngeal wall. Usually incidental finding on MRI. Smooth, well-circumscribed, cystic. | Can appear as a nasopharyngeal mass on imaging, but it is cystic (bright on T2 MRI), midline, and has no solid enhancement. Distinguished by imaging characteristics |
| Nasopharyngeal retention cyst | Small benign mucosal cyst from obstructed mucous glands. Very common incidental finding. | Smooth, small, cystic — clinically insignificant but must not be confused with a submucosal NPC |
| Condition | Key Distinguishing Features | Why it mimics NPC |
|---|---|---|
| Juvenile nasopharyngeal angiofibroma (JNA) [6] | Benign but locally aggressive vascular tumour of the nasopharynx. Occurs almost exclusively in adolescent males (10–25 years). Arises from the sphenopalatine foramen area. Presents with unilateral nasal obstruction and recurrent profuse epistaxis. Highly vascular — biopsy is contraindicated (risk of catastrophic haemorrhage). Diagnosis is clinical + imaging (CT/MRI showing Holman-Miller sign: anterior bowing of the posterior wall of the maxillary sinus). Characteristic intense contrast enhancement on CT/MRI. | Can present with similar nasal and otological symptoms to NPC (epistaxis, nasal obstruction, middle ear effusion from Eustachian tube compression). However, JNA occurs in young males, is highly vascular on imaging, and does NOT produce cranial nerve palsies or cervical lymphadenopathy (it is benign and does not metastasize) |
| Inverted papilloma [6] | Benign epithelial neoplasm of the sinonasal cavity. Arises from the lateral nasal wall (middle meatus typically), but can extend posteriorly into the nasopharynx. Associated with HPV. Has a ~10% risk of malignant transformation to SCC. Unilateral nasal obstruction, epistaxis. | Can present as a unilateral nasal/nasopharyngeal mass with similar symptoms to NPC. Distinguished by its typical lateral nasal wall origin, papillomatous appearance, and the absence of EBV association or cervical lymphadenopathy. Biopsy is definitive |
JNA vs NPC — Critical Distinction
Both JNA and NPC can present in the nasopharynx with epistaxis and nasal obstruction. However: JNA = adolescent male, no lymphadenopathy, intensely vascular (do NOT biopsy blindly — catastrophic bleed risk). NPC = middle-aged adult, cervical lymphadenopathy common, EBV-associated, biopsy is safe and diagnostic. Always get imaging before biopsy if JNA is suspected.
| Condition | Key Distinguishing Features | Why it mimics NPC |
|---|---|---|
| Non-Hodgkin lymphoma (NHL) [6][7] | The nasopharynx contains abundant lymphoid tissue (Waldeyer's ring) and is a recognized site for extranodal NHL. Tonsils and tongue base may be the presenting site for a lymphoma [7]. Nasopharyngeal lymphoma presents similarly to NPC: nasopharyngeal mass, nasal obstruction, cervical lymphadenopathy. However, lymphoma may cause diffuse symmetric enlargement of Waldeyer's ring tissue rather than a unilateral irregular mass. B symptoms (fever, night sweats, weight loss > 10%) more prominent. EBV-negative (usually). Diagnosis requires biopsy with immunohistochemistry and flow cytometry for subtyping — FNA alone is insufficient. In Hong Kong, NK/T-cell nasal-type lymphoma (extranodal NK/T-cell lymphoma) is a specific entity that involves the nasal cavity and nasopharynx, is EBV-associated, and has a destructive midline pattern | Overlapping presentation with NPC: nasopharyngeal mass, cervical lymphadenopathy, B symptoms. Key differentiators: lymphoma tends to be more symmetric, causes diffuse Waldeyer's ring enlargement, EBV status differs by subtype, and histology is lymphoid not epithelial. Biopsy with adequate tissue for IHC is essential — incisional biopsy or excisional biopsy may be needed since FNA does not provide material for tissue architecture or immunohistochemical analysis [8] |
| Sinonasal carcinoma [6] | Encompasses SCC, adenocarcinoma, sinonasal undifferentiated carcinoma (SNUC), adenoid cystic carcinoma. Risk factors include smoking and hard-wood exposure (for adenocarcinoma) [6]. Arises from the nasal cavity or paranasal sinuses and can extend posteriorly into the nasopharynx. Presents with unilateral obstruction, epistaxis, bleeding, cacosmia, proptosis, diplopia, epiphora, neurological symptoms [6]. | Posterior extension of a sinonasal tumour into the nasopharynx can mimic a primary NPC. Distinguished by the primary epicentre being in the nasal cavity/sinuses rather than the fossa of Rosenmüller, different histological subtypes, and different risk factor profile (wood dust, smoking rather than EBV). Imaging (CT/MRI) clarifies the epicentre |
| Minor salivary gland tumour [7] | May present as submucosal masses in the tongue base and soft palate [7], and can also arise in the nasopharyngeal mucosa (which contains minor salivary glands). Common malignant types: adenoid cystic carcinoma, mucoepidermoid carcinoma. Adenoid cystic carcinoma is notorious for perineural invasion (causes pain, numbness along nerve distribution). | Submucosal nasopharyngeal mass can be mistaken for NPC. However, minor salivary gland tumours have smooth overlying mucosa, are not EBV-associated, and have different histology (glandular, not squamous/undifferentiated). Biopsy is definitive |
| Malignant melanoma [6] | Rare. Sinonasal melanoma is an aggressive tumour. Can arise from melanocytes in the nasal or nasopharyngeal mucosa. Presents with epistaxis, obstruction. May appear pigmented (dark mass) or amelanotic. | Pigmented mucosal lesion is a clue but amelanotic variants can be deceptive. IHC stains (S100, HMB-45, Melan-A) are diagnostic |
| Olfactory neuroblastoma (esthesioneuroblastoma) [6] | Neural origin tumour arising from olfactory neuroepithelium at the cribriform plate. Can extend into nasopharynx. Presents with anosmia, epistaxis, nasal obstruction. | Superior nasal cavity mass extending posteroinferiorly. Distinguished by origin at cribriform plate and neuroendocrine histology |
| Sarcomas [6] | Mesenchymal malignancy. Rhabdomyosarcoma is the most common soft tissue sarcoma in children and can arise in the nasopharynx. In adults, consider leiomyosarcoma, fibrosarcoma. | Nasopharyngeal mass in a child → think rhabdomyosarcoma. Biopsy with IHC (desmin, myogenin) is diagnostic |
B. Differential Diagnosis When the Presenting Complaint is a Neck Mass
Since the most common initial presenting symptom of NPC is cervical lymphadenopathy [2], the differential diagnosis of a neck mass is crucial. The approach is organized by the clinical context.
When a patient presents with a neck mass, a systematic search for the primary malignancy is essential. This includes: right tonsillectomy and frozen section, left tonsillectomy, pharyngoscopy with biopsy of hypopharynx and tongue base, and nasopharyngoscopy and biopsy [9].
| Condition | Key Distinguishing Features | How to differentiate from NPC |
|---|---|---|
| Metastatic H&N SCC (non-NPC) [7] | Metastatic H&N cancer is predominantly related to metastatic squamous cell carcinoma arising from the aerodigestive tract [7]. Primary sites include oral cavity, oropharynx (HPV-associated), hypopharynx, larynx. Risk factors: smoking (primary risk factor), alcohol (synergistic effect), betel nut chewing (oral cavity), HPV (oropharyngeal) [4]. Usually presents in older patients (age > 60) [7]. Symptoms related to primary site: hoarseness (larynx), dysphagia (hypopharynx), oral ulcer (oral cavity). 90% of H&N malignancies are SCC (not including nasopharynx and thyroid) [4] | NPC is EBV-driven, presents in younger patients (40–60), has non-keratinizing histology, is radiosensitive. Other H&N SCCs are keratinizing, smoking/alcohol-driven, HPV-driven (oropharynx), and primary treatment often includes surgery. Nasopharyngoscopy with biopsy and EBV testing (EBER-ISH, plasma EBV DNA) differentiates |
| Oropharyngeal carcinoma [7][10] | Tonsil is the commonest sub-site [10]. Histology: SCC or lymphoma/minor salivary gland [10]. HPV-associated oropharyngeal carcinoma in young males with higher lifetime sexual partners [7]. Presents with sore throat, dysphagia, unilateral tonsillar enlargement, referred otalgia. Level II nodal metastasis common. FNA can be used for PCR testing — HPV for oropharyngeal SCC, EBV for NPC [8] | HPV+ oropharyngeal SCC is p16-positive on IHC, EBV-negative. NPC is EBV-positive, HPV-negative. Different treatment approaches (HPV+ oropharyngeal SCC may qualify for de-intensification) |
| Thyroid carcinoma [7] | Thyroid nodules confirmed with ultrasound and FNA [7]. Presents as anterior neck mass, usually lower neck (Level VI region). Well-differentiated thyroid cancers (papillary, follicular) are common and have excellent prognosis. | Location (anterior/central compartment), thyroid function tests, ultrasound characteristics, and FNA cytology (Bethesda system) distinguish from NPC nodes which are lateral level II |
| Lymphoma (Hodgkin and Non-Hodgkin) [7] | Neck involvement is common in children with Hodgkin lymphoma (80%) [7]. Presents with fever, night sweats, chills, and diffuse lymphadenopathy [7]. Rubbery, non-tender, mobile nodes. May have splenomegaly. Mediastinal lymphadenopathy on CXR (especially Hodgkin). | Lymphoma nodes are typically rubbery and mobile (NPC nodes may be hard and fixed). B symptoms are more prominent in lymphoma. Biopsy (excisional preferred for lymphoma subtyping) with IHC and flow cytometry is essential — FNA is insufficient for lymphoma subtyping [8] |
| Salivary gland tumour [7] | 80% of salivary tumours arise in the parotid gland. Parotid tumours are usually benign (80%). Submandibular gland tumours are usually malignant (50%) [7]. Presents as a parotid or submandibular swelling. Facial nerve palsy suggests parotid malignancy. | Location in the salivary gland region (parotid angle, submandibular triangle) is different from typical NPC level II nodes. FNA/core biopsy and imaging distinguish |
| Condition | Key Features | How to differentiate |
|---|---|---|
| Reactive lymphadenopathy | Common in young patients. Tender, mobile, often bilateral. History of recent URTI. Resolves spontaneously. | Short history, tenderness, resolution over weeks. NPC nodes are painless, firm, progressive |
| Tuberculous lymphadenitis (scrofula) | Important differential in Hong Kong (intermediate TB burden). Chronic non-tender cervical lymphadenopathy, often matted. May have systemic symptoms. Posterior triangle (Level V) predilection. Caseous necrosis on FNA/biopsy, AFB positive. | Can mimic NPC nodal disease. FNA showing granulomatous inflammation with caseation, positive AFB/TB PCR distinguishes. Always send FNA for TB culture in HK |
| Branchial cleft cyst [11] | Accounts for 20% of paediatric neck mass [11]. 2nd branchial cleft cyst is most common — presents inferior to angle of mandible and anterior to SCM [11]. Usually young patient, presents when cyst becomes infected. Cystic on USS/CT. | Cystic nature on imaging. Young patient. However, cystic metastatic NPC nodes or cystic metastatic HPV+ oropharyngeal SCC can mimic a branchial cyst in an older adult — any cystic lateral neck mass in a patient > 40 must be presumed malignant until proven otherwise |
| Paraganglioma (Carotid body tumour) [7] | Neoplasm arising from extra-adrenal chromaffin cells of the parasympathetic paraganglia. Highly vascular and typically benign [7]. Pulsatile, with bruit. Mobile side-to-side but not up-and-down (Fontaine's sign) [7]. Located at the carotid bifurcation. | Pulsatile nature and Fontaine's sign are pathognomonic. Contrast CT/MRA shows intensely enhancing mass at carotid bifurcation ("splaying" of ICA and ECA). Do NOT biopsy (highly vascular) |
| Schwannoma [7] | Neoplastic proliferation of Schwann cells. Commonly arises from the vagus nerve or superior cervical sympathetic chain [7]. Vagal schwannoma may cause hoarseness; sympathetic chain schwannoma may cause Horner syndrome [7]. Slow-growing, well-encapsulated. | Slow growth, specific nerve-related symptoms, well-defined encapsulated mass on MRI (target sign). No EBV association |
| Lipoma / Benign skin cyst [7] | Soft, ill-defined, slowly enlarging mass [7]. Epidermoid cyst, dermoid, pilomatrixoma. | Soft consistency, superficial location, non-fixed |
The 'Cystic Neck Mass' Trap
A common clinical pitfall: a cystic lateral neck mass in an adult > 40 years is NOT a branchial cleft cyst until proven otherwise. Cystic metastatic nodes from NPC or HPV-positive oropharyngeal SCC are a far more common diagnosis in this age group. Always investigate further with imaging, FNA (for cytology + EBV/HPV PCR), and nasopharyngoscopy.
Sometimes the patient does not present with a neck mass but with one of the other clinical features of NPC. The DDx then shifts:
| Presenting Symptom | DDx (besides NPC) | Rationale |
|---|---|---|
| Unilateral middle ear effusion in an adult | NPC (must exclude first), nasopharyngeal lymphoma, other nasopharyngeal tumour, idiopathic Eustachian tube dysfunction, radiation fibrosis (post-RT), cleft palate (rare in adults) | Any mass obstructing the Eustachian tube can cause effusion. In endemic areas, NPC is the most important cause to rule out |
| Unilateral epistaxis + nasal obstruction | NPC, sinonasal carcinoma, inverted papilloma, JNA (adolescent males), nasal polyps, foreign body, hereditary haemorrhagic telangiectasia. Red flags for sinonasal neoplasm: unilateral obstruction, epistaxis, bleeding, cacosmia, proptosis, diplopia, epiphora, neurological symptoms [6] | Bilateral symptoms are more likely benign (polyps, rhinitis). Unilateral nasal symptoms are a red flag for neoplasm [6] |
| Cranial nerve palsy (especially CN V, VI) | NPC (skull base invasion), skull base meningioma, chordoma (clivus), nasopharyngeal lymphoma, perineural spread of adenoid cystic carcinoma, cavernous sinus thrombosis, pituitary apoplexy, Tolosa-Hunt syndrome | Skull base imaging (MRI with gadolinium) is essential. NPC is distinguished by nasopharyngeal mass + EBV positivity |
| Bilateral cervical lymphadenopathy | NPC (midline tumour → bilateral drainage), lymphoma, TB, sarcoidosis, HIV, metastatic disease from other primaries | NPC is one of the few H&N cancers that commonly causes bilateral cervical nodes because it is a midline structure |
When a patient presents with a metastatic cervical lymph node and no obvious primary tumour on initial examination, a systematic search protocol is essential:
The search for the primary malignancy includes [9]:
- Nasopharyngoscopy and biopsy — to look for NPC [9]
- Right tonsillectomy and frozen section [9] — HPV+ oropharyngeal SCC often presents as a tiny or submucosal tonsillar primary
- Left tonsillectomy [9]
- Pharyngoscopy with biopsy of hypopharynx and tongue base [9]
- Panendoscopy (direct laryngoscopy, bronchoscopy, OGD) [3] — to exclude synchronous primary
Additionally:
- FNA of the neck node with cytology, EBV PCR (for NPC) and HPV PCR (for oropharyngeal SCC) [8]
- Plasma EBV DNA — if elevated, strongly points to NPC as the occult primary
- PET-CT — to identify the primary site and detect distant metastases
High Yield: The FNA of a cervical metastatic node can direct you to the primary: EBV-positive → think NPC; HPV/p16-positive → think oropharyngeal SCC. This avoids unnecessary exploration and guides the endoscopic search.
| Feature | NPC | NHL (nasopharyngeal) | JNA | Sinonasal carcinoma | HPV+ oropharyngeal SCC |
|---|---|---|---|---|---|
| Age/Sex | 40–60, M > F | Any age | Adolescent male | Older adults | Younger males (30–50) |
| EBV | Positive (virtually 100% endemic) | Variable (NK/T-cell = EBV+) | Negative | Negative | Negative |
| HPV | Negative | Negative | Negative | Negative | Positive (HPV 16/18) |
| Biopsy safe? | Yes | Yes (need adequate tissue for IHC) | NO — risk of massive haemorrhage | Yes | Yes |
| Cervical LN | Very common, bilateral | Common | Absent | Less common | Common (cystic nodes) |
| Radiosensitive | Very radiosensitive | Variable by subtype | Not radiosensitive (surgery) | Moderate | Very radiosensitive |
| Key investigation | EBER-ISH, plasma EBV DNA | IHC, flow cytometry | CT/MRI angiography | CT for bone, MRI for soft tissue | p16 IHC, HPV ISH |
High Yield Summary — Differential Diagnosis of NPC
Nasopharyngeal mass DDx: NPC (most common malignancy), NHL (especially NK/T-cell lymphoma in HK), JNA (adolescent males — DO NOT biopsy), sinonasal carcinoma extending posteriorly, minor salivary gland tumour, melanoma, rhabdomyosarcoma (children).
Neck mass DDx: Metastatic NPC, other metastatic H&N SCC (oropharyngeal HPV+, hypopharyngeal, laryngeal), lymphoma (Hodgkin/NHL), thyroid carcinoma, TB lymphadenitis, branchial cleft cyst (beware in adults > 40 — likely cystic met), paraganglioma, schwannoma.
Key differentiating tools: FNA with EBV PCR (NPC) vs HPV PCR (oropharyngeal SCC); EBER-ISH on biopsy; plasma EBV DNA; p16 IHC; imaging characteristics (JNA = intensely vascular, do not biopsy).
Search for occult primary: Nasopharyngoscopy + bilateral tonsillectomy + tongue base/hypopharynx biopsy + panendoscopy. FNA of node with EBV/HPV PCR guides the search.
Red flag: Any unilateral nasal symptom or adult unilateral middle ear effusion → exclude nasopharyngeal neoplasm.
Active Recall - Differential Diagnosis of NPC
References
[1] Lecture slides: GC 215. Common nasal conditions and nasopharyngeal carcinoma (1).pdf (p47–48) [2] Senior notes: felixlai.md (felix:357, 364) — Nasopharyngeal cancer (NPC) [3] Senior notes: felixlai.md (felix:342) — Head and neck cancer overview, panendoscopy [4] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p40–41) [6] Lecture slides: GC 215. Common nasal conditions and nasopharyngeal carcinoma (1).pdf (p41–44) [7] Senior notes: felixlai.md (felix:299, 369) — Differential diagnosis of neck mass, oropharyngeal carcinoma etiology [8] Senior notes: felixlai.md (felix:303) — Pathological tests (FNA, biopsy) [9] Lecture slides: GC 218. I have a swelling in the neck Neck mass.pdf (p9) [10] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p36) [11] Senior notes: felixlai.md (felix:295) — Branchial cleft cyst
Diagnosis of Nasopharyngeal Carcinoma
NPC diagnosis rests on three pillars: (1) tissue diagnosis (histopathological confirmation via endoscope-guided biopsy), (2) EBV biomarkers (serology and plasma EBV DNA), and (3) imaging (for staging and assessment of disease extent). Let me walk you through each in a logical, clinical order — the way you would actually work up a patient in Hong Kong.
The core concept: you cannot stage what you haven't diagnosed, and you cannot treat what you haven't staged. The diagnostic algorithm therefore follows: suspicion → endoscopy with biopsy → confirm histology and EBV status → stage with imaging → plan treatment.
There is no single "diagnostic criteria checklist" for NPC like there is for, say, rheumatoid arthritis. Instead, the definitive diagnosis requires histopathological confirmation from a biopsy of the primary nasopharyngeal tumour. However, the clinical pathway integrates several elements:
| Component | Requirement | Rationale |
|---|---|---|
| Histopathological confirmation | Endoscope-guided biopsy of the primary tumour [2] | This is the gold standard. Tissue provides WHO classification, differentiation status, and material for EBER in situ hybridization |
| EBV confirmation | EBER-ISH positive on tumour tissue (for non-keratinizing NPC in endemic areas) | Virtually 100% of non-keratinizing NPC is EBER-positive. Confirms EBV causation and distinguishes from other nasopharyngeal malignancies |
| Supporting biomarkers | Plasma EBV DNA (quantitative PCR) [2]; EBV VCA-IgA and EA-IgA serology [2] | Supports diagnosis, aids staging/prognostication, and provides baseline for monitoring |
| Staging investigations | MRI nasopharynx/skull base/neck + PET-CT or CT chest/abdomen + bone scan | Determines TNM stage which dictates treatment approach |
Key Principle
Definitive diagnosis is made by endoscope-guided biopsy of the primary tumour [2]. You must see the tumour with an endoscope and biopsy it. Incisional nodal biopsy or nodal dissection should be avoided since it will negatively impact subsequent treatment [2] — because NPC is treated primarily with radiation-based therapy, not surgery. Violating the neck surgically can compromise radiation fields, cause scarring that alters lymphatic drainage, and seed tumour cells.
The following algorithm represents the clinical pathway from initial suspicion to definitive staging:
3. Investigation Modalities — Detailed Breakdown
A. Endoscopic Examination
- What it is: Flexible fibreoptic nasopharyngoscopy (passed through the nose) or rigid endoscopy. Allows direct visualization of the nasopharynx including the fossa of Rosenmüller, torus tubarius, nasopharyngeal roof, and posterior choanae.
- Why it is the first investigation: The nasopharynx is a clinically occult site [2] — you cannot see it with the naked eye. Indirect mirror examination (posterior rhinoscopy) is possible but suboptimal. Flexible endoscopy is the standard of care.
- Key findings:
- Exophytic mass arising from the fossa of Rosenmüller — irregular, friable, may be ulcerated
- Submucosal bulge — NPC can grow submucosally, making the mucosa look deceptively normal with just a bulge
- Mucosal irregularity or asymmetry — even subtle asymmetry of the fossa of Rosenmüller warrants biopsy
- Blood or crusting at the tumour surface
- Biopsy: Endoscope-guided biopsy of the primary tumour provides definitive diagnosis [2]. Multiple biopsies should be taken (at least 3–4 pieces). If no obvious mass is seen but clinical suspicion is high (e.g., EBV DNA positive, cervical metastatic node), random biopsies of both fossae of Rosenmüller and the nasopharyngeal roof should be performed.
- Panendoscopy includes direct laryngoscopy, bronchoscopy, and OGD [3]
- 10% risk of synchronous/metachronous tumour due to field cancerization [12]
- Staging examination is recommended at the initial evaluation of all patients with primary cancers of the upper aerodigestive tract [3]
- While field cancerization is more relevant for smoking/alcohol-driven H&N cancers than for EBV-driven NPC, panendoscopy is still part of the standard workup to exclude a second primary
Why not just biopsy the neck node?
Students sometimes ask: "If the patient has a big neck node, why not just excise it?" The answer: incisional nodal biopsy or nodal dissection should be avoided since it will negatively impact subsequent treatment [2]. NPC is treated with radiotherapy ± chemotherapy. Surgical violation of the neck can cause scarring, alter lymphatic drainage patterns, compromise radiation planning, and potentially seed tumour cells into the surgical bed. Instead, do FNA of the neck node (for cytology + EBV PCR) and biopsy the primary in the nasopharynx.
B. Laboratory Investigations
- What it is: Quantitative real-time PCR measuring circulating EBV DNA fragments in plasma. These DNA fragments are released from NPC tumour cells undergoing apoptosis and necrosis.
- Why it is useful: This is arguably the most important single biomarker in NPC management. It serves four roles:
| Role | Explanation |
|---|---|
| Screening | Hong Kong studies (Prof Dennis Lo, CUHK) demonstrated that a single plasma EBV DNA test can screen asymptomatic individuals for early-stage NPC. A positive result triggers nasopharyngoscopy and MRI. Significantly shifts stage distribution toward earlier detection |
| Diagnostic and staging evaluation for prognosis [2] | Pre-treatment plasma EBV DNA levels correlate with outcomes [2] — higher levels = larger tumour burden = worse stage = worse prognosis |
| Treatment response [2] | Post-treatment plasma EBV DNA levels evaluate treatment response and detection of recurrence [2]. After successful treatment, EBV DNA should become undetectable. Persistent or rising levels after treatment indicate residual/recurrent disease |
| Surveillance | Serial measurements during follow-up detect relapse earlier than clinical symptoms or imaging |
- Key findings and interpretation:
- Detectable pre-treatment level → supports NPC diagnosis and indicates active disease
- Level correlates with tumour stage → higher copy number = higher stage
- Undetectable post-treatment → good response
- Detectable/rising post-treatment → relapse (PET-CT needed to locate recurrence)
High Yield: Plasma EBV DNA is both the most sensitive and most specific circulating biomarker for NPC in endemic populations. It has largely superseded EBV serology for clinical use.
- Sustained rise in IgA to both viral capsid antigen (VCA) and early antigen (EA) [2]
- ↑ EBV VCA-IgA and ↑ EBV EA-IgA [2]
- Why IgA? The nasopharynx is a mucosal surface. The dominant immunoglobulin at mucosal surfaces is IgA (secretory immune system). NPC triggers a mucosal immune response → IgA class antibodies against EBV antigens are produced locally and enter the bloodstream.
- EBV-specific serological screening has low specificity for NPC [2] — because > 90% of the population is EBV-seropositive (IgG), and even VCA-IgA can be transiently elevated during EBV reactivation in healthy individuals. Hence, serology has high sensitivity but low specificity for NPC, leading to false positives.
- Elevated titre may precede diagnosis of NPC for a period of up to 10 years [2] — this long lead time supports the concept of using serology for population screening, but the low specificity is a limitation.
- Practical role: Serology is used as a population screening tool (especially VCA-IgA combined with EA-IgA), but plasma EBV DNA is now preferred for its superior specificity.
- Why: Baseline haematological assessment. Can reveal anaemia of chronic disease (advanced malignancy), cytopenias if bone marrow infiltrated (rare, advanced M1 disease), or lymphocytosis if considering lymphoma differential.
- ↑ ALP level in bone metastasis [2]
- Why: Bone is the most common site of distant metastasis in NPC (~75%). Osteoblastic or mixed bone metastases increase osteoblast activity → elevated alkaline phosphatase (bone isoform). However, ALP is not specific — can be elevated by liver disease, other bone pathology, or even normal growth in adolescents. It serves as a screening blood test to raise suspicion for bone metastasis, prompting further imaging.
| Test | Rationale |
|---|---|
| Liver function tests (LFT) | Baseline for chemotherapy planning; elevated transaminases/GGT may suggest liver metastasis |
| Renal function tests (RFT) | Baseline before cisplatin-based chemotherapy (cisplatin is nephrotoxic) |
| Electrolytes | Pre-chemotherapy baseline |
| Lactate dehydrogenase (LDH) | Non-specific tumour marker; elevated in high tumour burden |
C. Pathological Tests
- When used: When the patient presents with a neck mass and the primary site is not yet known, FNA of the cervical lymph node is the initial pathological investigation.
- Aspirate is used for cytological analysis, PCR testing for virus [8]:
- FNA does NOT provide material for tissue architecture or immunohistochemical analysis [8] — this is a critical limitation. FNA gives you individual cells (cytology), not tissue blocks. You cannot assess tumour architecture, perform comprehensive IHC panels, or do flow cytometry on FNA alone. This matters particularly when lymphoma is in the differential.
- Interpretation:
- FNA showing malignant squamous/undifferentiated epithelial cells + EBV PCR positive → strongly suggests NPC → proceed to nasopharyngoscopy and biopsy of primary
- FNA showing lymphoid cells → suspect lymphoma → excisional biopsy needed for subtyping [8]
- Gold standard for definitive diagnosis
- Tissue submitted for:
- H&E histopathology: Determines WHO type (non-keratinizing undifferentiated/differentiated, keratinizing SCC, basaloid SCC)
- EBER in situ hybridization (EBER-ISH): Detects EBV-encoded small RNAs in tumour cell nuclei. This is the gold standard for demonstrating EBV in tumour tissue. Positive nuclear staining confirms EBV association.
- Immunohistochemistry (IHC): Cytokeratin positivity (AE1/AE3, CK5/6) confirms epithelial origin — distinguishes NPC from lymphoma. p63 often positive. For lymphoepithelial carcinoma of salivary glands: histology is exactly like NPC, EBV-related, tumour cells EBER-positive [13]
- p16 IHC / HPV ISH: If keratinizing SCC pattern and clinical suspicion of oropharyngeal origin (NPC vs extension from oropharynx)
- Incisional nodal biopsy or nodal dissection should be avoided [2] in the setting of suspected NPC
- However, biopsy of a cervical node IS appropriate when:
- Lymphoma is suspected (FNA inconclusive) — excisional biopsy is considered when FNA or core needle biopsy is positive for lymphoma where additional tissue is required for subtyping [8]
- The primary site genuinely cannot be identified despite full workup
D. Imaging Investigations
-
Why MRI and not CT for the primary?: MRI provides optimal visualization of soft-tissue infiltration of the tumour [14]. The nasopharynx is surrounded by critical soft tissue structures (parapharyngeal fat, muscles, cavernous sinus, brain). MRI's superior soft tissue contrast allows precise delineation of:
- Extent of primary tumour (T staging)
- Parapharyngeal space invasion
- Skull base involvement (cortical bone erosion, marrow infiltration)
- Cavernous sinus invasion
- Intracranial extension
- Retropharyngeal and cervical lymph node metastases (N staging)
- Perineural spread (enhancement along CN V, seen as thickening and enhancement of the nerve on post-gadolinium fat-suppressed T1)
-
Key MRI sequences and findings:
| Sequence | Finding in NPC |
|---|---|
| T1-weighted | Tumour is isointense to slightly hypointense relative to muscle. Loss of normal parapharyngeal fat plane (bright on T1) indicates lateral invasion |
| T2-weighted | Tumour is intermediate-to-high signal. Helps delineate tumour from surrounding structures |
| Post-gadolinium T1 with fat suppression | Tumour enhances avidly. This is the most important sequence for delineating tumour extent, skull base invasion, cavernous sinus involvement, and perineural spread |
| DWI (Diffusion-Weighted Imaging) | Restricted diffusion (bright on DWI, dark on ADC map) in tumour — reflects high cellularity. Useful for distinguishing tumour from post-treatment changes |
- Assessment of locoregional disease extent [2] — MRI is essential for accurate T staging
- Useful to detect bony invasion [14] — CT has superior spatial resolution for cortical bone compared to MRI
- Detection of cervical lymph node metastasis [14]
- CT thorax and abdomen to assess for distant metastasis [14]
- CT of the nasopharynx/skull base is complementary to MRI (bone windows show cortical erosion at skull base, pterygoid plates, etc.)
- Contrast-enhanced CT neck: Shows enhancing primary tumour and lymph nodes. Necrotic nodes (central low-attenuation with rim enhancement) suggest metastatic disease.
- Uses 18-fluorodeoxyglucose (18F-FDG) as tracer [2] (note: the senior notes say "17-FDG" which is a typo — the correct isotope is 18F-FDG)
- How it works: 18F-FDG is a glucose analogue taken up by metabolically active cells. Cancer cells have upregulated glucose transporters (GLUT-1) and hexokinase → trap FDG intracellularly → appear as "hot spots" on PET
- Superior ability to detect lymph node and bone metastasis [2]
- Key roles in NPC:
| Role | Explanation |
|---|---|
| Distant metastasis detection (M staging) | PET-CT is the most sensitive single investigation for detecting distant metastases (bone, liver, lung, distant nodes). This is its primary role in NPC staging |
| Nodal assessment | Can detect metabolically active nodes that appear normal in size on CT/MRI |
| Identifying occult primary | When a patient presents with a metastatic cervical node and no visible primary on endoscopy, PET-CT can localize the primary |
| Treatment response assessment | Post-treatment PET-CT can distinguish residual active tumour from post-radiation fibrosis/inflammation (though timing is important — usually done ≥ 12 weeks post-treatment to reduce false positives) |
| Recurrence detection | Complements plasma EBV DNA for localizing recurrent disease |
- Interpretation:
- SUVmax (Standardised Uptake Value) > 2.5 generally suggests malignancy
- False positives: infection, inflammation, granulomatous disease (TB — important in HK)
- False negatives: small lesions ( < 1 cm), low-grade tumours (NPC is usually FDG-avid, so this is rarely an issue)
- Evaluate for lung metastasis [2]
- Why: Simple, quick, cheap. Detects pulmonary metastases (nodules, pleural effusion). However, sensitivity is limited compared to CT chest. Often used as initial screen; if suspicious or if PET-CT is not available, CT chest is performed.
- Evaluate for bone metastasis [2]
- How it works: 99mTc-MDP (methylene diphosphonate) is a bone-seeking radiopharmaceutical that accumulates in areas of increased osteoblastic activity. Metastatic bone disease triggers a reparative osteoblastic response → increased tracer uptake → "hot spots"
- Why used: Bone is the most common site of distant metastasis in NPC (75%). Bone scan surveys the entire skeleton in one study.
- Limitations: Not specific — also picks up fractures, degenerative disease, Paget's disease. Purely lytic metastases (minimal osteoblastic response) may be missed. Largely being replaced by PET-CT which is more sensitive and specific.
- Ultrasound neck +/- FNAC [12] — used for assessment of cervical lymph nodes
- Role in NPC: Can assess size, morphology, and vascularity of cervical lymph nodes. Ultrasound features suggesting malignancy: rounded shape (L/S ratio < 2), loss of fatty hilum, irregular cortical thickening, peripheral/chaotic vascularity, necrosis. Can guide FNA of suspicious or equivocal nodes.
| Investigation | Purpose | Key Findings in NPC |
|---|---|---|
| Nasopharyngoscopy + biopsy | Definitive tissue diagnosis | Mass at fossa of Rosenmüller; biopsy → non-keratinizing undifferentiated carcinoma, EBER-ISH positive |
| Plasma EBV DNA | Diagnosis, staging, prognosis, monitoring | Detectable and quantifiable; correlates with tumour burden; undetectable post-treatment = good response |
| EBV VCA-IgA, EA-IgA | Screening, supportive diagnosis | Elevated; low specificity; may precede diagnosis by up to 10 years |
| CBC, ALP, LFT, RFT | Baseline bloods, metastasis screening | ↑ ALP suggests bone mets; ↑ LFT suggests liver mets; baseline for chemotherapy |
| FNA of neck node | Initial pathological assessment of neck mass | Malignant cells + EBV PCR positive → suspect NPC; guides search for primary |
| MRI nasopharynx/skull base/neck | T and N staging, soft tissue assessment | Tumour extent, parapharyngeal invasion, skull base erosion, cavernous sinus involvement, retropharyngeal and cervical nodes |
| CT | Bony invasion assessment, distant metastasis | Skull base bone erosion; CT chest/abdomen for lung/liver mets |
| PET-CT | M staging, occult primary detection | FDG-avid primary and metastatic sites; superior for bone and distant nodal mets |
| CXR | Lung metastasis screening | Pulmonary nodules, pleural effusion |
| Bone scan | Bone metastasis screening | Hot spots at common sites (spine, pelvis, ribs) |
| Panendoscopy | Exclude synchronous primary | Direct laryngoscopy + bronchoscopy + OGD |
Once NPC is histologically confirmed, the staging workup determines the TNM stage which dictates treatment:
| Stage Component | Investigation | What You're Looking For |
|---|---|---|
| T stage | MRI nasopharynx and skull base | Primary tumour extent: confined to nasopharynx (T1), parapharyngeal extension (T2), skull base/sinuses (T3), intracranial/CN/orbit (T4) |
| N stage | MRI neck + PET-CT | Retropharyngeal nodes, unilateral vs bilateral cervical nodes, size relative to 6 cm, position relative to caudal border of cricoid |
| M stage | PET-CT (or CT chest/abdomen + bone scan) | Bone, liver, lung, distant nodal metastases |
| Biomarker | Plasma EBV DNA (quantitative) | Tumour burden, prognostic stratification |
The rationale for screening [2]:
- High rate of NPC in specific geographical regions and among certain families [2]
- High rate of cure for early-stage NPC [2] — 5-year overall survival for Stage I NPC is > 90%; for Stage IVB it drops to ~50–60%. Early detection dramatically improves outcomes.
Two practical ways to screen for EBV [2]:
- Immunoglobulin A viral capsid antigen (EBV VCA-IgA) [2] — traditional screening method, but limited specificity
- Plasma EBV DNA by PCR [2] — newer, more specific approach
The Hong Kong NPC Screening Study (2017, NEJM — Prof Dennis Lo, CUHK):
- Prospective screening of > 20,000 asymptomatic men aged 40–62 using plasma EBV DNA
- Two-time-point approach: initial test → if positive, repeat in 4 weeks → if persistently positive, nasopharyngoscopy + MRI
- Results: detected NPC at significantly earlier stages (71% Stage I/II in screened group vs ~20% in symptom-detected population)
- Landmark study supporting population-based screening in high-risk populations
Screening Algorithm
Step 1: Plasma EBV DNA (quantitative PCR) → Step 2: If positive, repeat in 4 weeks → Step 3: If persistently positive, nasopharyngoscopy + MRI nasopharynx → Step 4: Biopsy if mass seen; random biopsy + close surveillance if no mass. This two-time-point approach minimizes false positives from transient EBV reactivation.
EARLY REFERRAL to ENT surgeons when suspecting malignancy [17]:
- Persistent 2–4 weeks after conservative/empirical treatment [17]
- Clinically suspicious: irregular, induration, > 2 cm, associated cervical LN enlargement [17]
In the context of NPC specifically:
- Any adult with unilateral serous otitis media in an endemic area → scope the nasopharynx
- Any neck mass persistent > 2 weeks without obvious infective cause → investigate
- Any unilateral nasal symptoms (obstruction, epistaxis) in an adult → exclude neoplasm
- Any cranial nerve palsy without clear explanation → image the skull base
High Yield Summary — Diagnosis of NPC
Definitive diagnosis: Endoscope-guided biopsy of the nasopharyngeal primary → histopathology (WHO classification) + EBER-ISH (EBV confirmation).
Do NOT excise/biopsy neck nodes — NPC is treated with RT/CRT, not surgery. Nodal surgery compromises treatment.
Key biomarkers:
- Plasma EBV DNA (quantitative PCR) — diagnosis, staging, prognosis, treatment monitoring, recurrence detection. The single most important biomarker.
- EBV VCA-IgA and EA-IgA — screening use but low specificity. Titre may precede diagnosis by up to 10 years.
- ALP — elevated suggests bone metastasis.
FNA of neck node: Cytology + EBV PCR (for NPC) / HPV PCR (for oropharyngeal SCC). Guides search for primary. Does NOT provide tissue architecture.
Imaging:
- MRI = primary modality for T and N staging (superior soft tissue contrast).
- PET-CT = primary modality for M staging (superior for bone and distant metastases).
- CT = complementary for bone erosion; CT chest/abdomen if PET-CT unavailable.
- Bone scan / CXR = adjuncts if PET-CT unavailable.
Panendoscopy = exclude synchronous primary (10% risk from field cancerization).
Screening: Plasma EBV DNA (two-time-point approach) in high-risk populations → nasopharyngoscopy + MRI if persistently positive.
Active Recall - Diagnosis of Nasopharyngeal Carcinoma
References
[2] Senior notes: felixlai.md (felix:357, 364, 366) — NPC overview, diagnosis, screening [3] Senior notes: felixlai.md (felix:342, 369) — H&N cancer overview, panendoscopy [8] Senior notes: felixlai.md (felix:303) — Pathological tests (FNA, biopsy) [12] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p42) — Workup and investigation [13] Lecture slides: GC 217. Facial nerve palsy and salivary gland diseases.pdf (p76) — Lymphoepithelial carcinoma [14] Senior notes: felixlai.md (felix:371) — Diagnosis of oropharyngeal carcinoma (CT/MRI roles) [15] Lecture slides: GC 218. I have a swelling in the neck Neck mass.pdf (p7) — Investigations for neck mass [16] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p43) — Management framework [17] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p48) — Take home message, early referral
Management of Nasopharyngeal Carcinoma
Before diving into the treatment algorithm, you need to understand why NPC is the great exception among head and neck cancers. For virtually every other H&N cancer (oral cavity, oropharynx, hypopharynx, larynx), surgery is a first-line option — either alone for early stage or combined with adjuvant RT ± chemotherapy for late stage. NPC breaks this rule completely.
Three reasons NPC is treated with radiation-based therapy, not surgery:
- Deep anatomical location of the nasopharynx [2] — the nasopharynx sits deep in the skull, behind the nasal cavity, surrounded by the skull base superiorly, the carotid sheath and parapharyngeal space laterally, and the prevertebral fascia posteriorly. Surgical access is extremely difficult and would require morbid approaches with unacceptable functional consequences.
- Close proximity to critical neurovascular structures [2] — the internal carotid artery, cavernous sinus, and cranial nerves III–VI and IX–XII are all immediately adjacent. Surgical resection with adequate margins would risk devastating neurological deficits.
- NPC is exquisitely radiosensitive — particularly the non-keratinizing undifferentiated subtype (the endemic form in Hong Kong). This means radiation therapy achieves excellent local control rates without the morbidity of surgery.
The key lecture slide distinction [16]:
General rule for H&N cancer:
- Early stage: radiotherapy or minimally invasive surgery (laser/robotic) [16]
- Late stage: surgery with adjuvant treatment [16]
BUT — two critical exceptions:
This means NPC flips the usual paradigm: even in late-stage disease, you do NOT operate. You use chemo-irradiation.
Overview Table
Management according to stages [2]:
| Stage | Approach | Rationale |
|---|---|---|
| Early Stage I | Radiotherapy ONLY [2] | T1N0M0 — small tumour confined to nasopharynx with no nodal or distant disease. RT alone achieves > 90% 5-year overall survival. Adding chemotherapy provides no additional benefit and only adds toxicity |
| Intermediate Stage II | Concurrent chemoradiotherapy [2] | Parapharyngeal extension and/or N1 disease. Chemo (cisplatin) radiosensitizes the tumour — adding cisplatin during RT improves local control and overall survival compared to RT alone |
| Advanced Stage III | Concurrent chemoradiotherapy ± adjuvant chemotherapy [2] | Bulky nodal disease (N2) or skull base/sinus involvement (T3). Higher tumour burden warrants more aggressive systemic therapy |
| Stage IVA | Concurrent chemoradiotherapy ± adjuvant chemotherapy [2] | T4 or large nodes. Intracranial extension, CN involvement, orbit involvement |
| Stage IVB | Concurrent chemoradiotherapy + adjuvant chemotherapy [2] | N3 disease (nodes > 6 cm or below cricoid). High risk of distant failure |
| Stage IVC | Palliative systemic therapy | M1 disease (distant metastases). Cure is unlikely; goal shifts to prolongation of survival and symptom control |
The Big Picture — Treatment Escalation by Stage
Think of it as a stepwise escalation:
- Stage I → RT alone (single modality, low toxicity)
- Stage II–IVB → RT + cisplatin concurrently ± additional chemotherapy (combined modality, increasing intensity)
- Stage IVC → systemic chemotherapy ± palliative RT (palliation, not cure)
NPC management is always based on TNM staging [16]. Early stage = single modality; late stage = combined modality [16].
Treatment Modalities in Detail
1. Radiotherapy (RT)
- Remains the mainstay of treatment because NPC is a radiosensitive tumour and its anatomical location limits a surgical approach [2]
- Bilateral neck irradiation is always included since NPC has a propensity to spread to bilateral regional lymph nodes in the neck [2]
- Even in clinically N0 disease, both sides of the neck are irradiated because of the high rate of occult bilateral nodal micrometastases
- IMRT is the standard of care for NPC (replaced older 2D and 3D conformal techniques)
- Why IMRT? The nasopharynx is surrounded by dose-limiting critical structures: brainstem, spinal cord, temporal lobes, optic chiasm, parotid glands, cochlea, TMJ. IMRT uses computer-optimised modulated radiation beams to conform the high-dose volume tightly to the tumour while sparing surrounding normal structures
- Dose: Typically 70 Gy to the primary tumour and gross nodal disease (given in 33–35 fractions over ~7 weeks), with lower doses (50–60 Gy) to elective nodal regions
- IMRT has significantly reduced the incidence of xerostomia (dry mouth) compared to older techniques because it spares contralateral parotid gland tissue
| Setting | Details |
|---|---|
| Definitive RT alone | Stage I NPC (T1N0M0). Curative intent. > 90% 5-year OS |
| Concurrent with chemotherapy | Stage II–IVB NPC. Cisplatin given concurrently enhances radiosensitivity |
| Adjuvant RT | NOT typically applicable in NPC (RT is the primary, not adjuvant modality) |
| Palliative RT | Stage IVC — RT to primary for symptom control (e.g., bleeding, obstruction, pain) |
| Re-irradiation | For local recurrence — technically challenging due to cumulative normal tissue doses |
| Side Effect | Mechanism | Timing |
|---|---|---|
| Mouth and throat sores (mucositis) [2] | Radiation damages rapidly dividing mucosal epithelial cells → ulceration and inflammation | Acute (during and weeks after RT) |
| Xerostomia (dry mouth) | Radiation damages salivary gland acinar cells (serous acini are more radiosensitive than mucous) → reduced saliva production | Acute and chronic. IMRT reduces this |
| Dysphagia / Odynophagia | Pharyngeal mucosal inflammation and oedema; late fibrosis of pharyngeal constrictors | Acute and chronic |
| Dermatitis | Radiation damage to skin in the treatment field | Acute |
| Sensorineural hearing loss | Radiation to cochlea (inner ear is within the treatment field) | Chronic (months to years) |
| Trismus | Fibrosis of masticator muscles (pterygoids, masseter) and TMJ | Chronic |
| Hypothyroidism | Radiation to thyroid gland in the neck fields | Chronic (requires thyroid function monitoring) |
| Osteoradionecrosis of mandible | Radiation damages osteocytes and periosteal blood supply → avascular necrosis | Late (months to years). Risk increased by dental extraction post-RT |
| Temporal lobe necrosis | Radiation to temporal lobes (adjacent to nasopharynx) → white matter necrosis | Late (years). Potentially devastating. IMRT reduces incidence |
| Cranial neuropathy | Radiation-induced fibrosis/damage to cranial nerves | Late |
| Secondary malignancy | Radiation-induced DNA damage in normal tissues → second cancer in irradiated field | Very late (decades) |
Pre-RT Dental Assessment
All NPC patients should have a comprehensive dental assessment and any necessary extractions BEFORE starting RT. Dental extraction after RT carries a high risk of osteoradionecrosis because irradiated bone has compromised vascularity and healing capacity. This is a key clinical practice point.
2. Chemotherapy
Chemotherapy in NPC is used in several settings:
| Setting | Timing | Drugs | Rationale |
|---|---|---|---|
| Concurrent | Given simultaneously with RT | Cisplatin (single agent, weekly or 3-weekly) | Radiosensitization — cisplatin enhances radiation-induced DNA damage by inhibiting DNA repair. This is the backbone of NPC treatment for Stage II+ |
| Induction (neoadjuvant) | Given BEFORE concurrent CRT | Gemcitabine + Cisplatin (GP); or Docetaxel + Cisplatin + 5-FU (TPF) | Aims to reduce tumour bulk before RT and eradicate micrometastases early. Recent trials (e.g., JUPITER-02 and CAPTAIN-1st era studies, and the landmark 2019 SunYatSen GP induction trial) show induction GP + concurrent CRT improves overall survival compared to concurrent CRT alone in locoregionally advanced NPC |
| Adjuvant | Given AFTER completion of concurrent CRT | Cisplatin + 5-FU (PF); or guided by post-treatment plasma EBV DNA | Post-radiotherapy plasma EBV DNA is validated as the MOST significant prognostic biomarker to select high-risk patients for adjuvant chemotherapy [2]. Patients with detectable post-treatment EBV DNA are at high risk of relapse and may benefit from adjuvant chemotherapy |
| Palliative | Stage IVC (distant metastases) or recurrent disease | Cisplatin + Gemcitabine ± immunotherapy; or Carboplatin-based if cisplatin-ineligible | Aim is disease control and palliation, not cure |
- Alkylating agent [2] (technically a platinum-based coordination compound, but functionally acts like an alkylating agent)
- Think of it as a "molecular stapler" — it cross-links the two strands of DNA together so they cannot separate for replication or transcription
Why cisplatin is ideal for concurrent CRT: Cisplatin's DNA cross-linking inhibits the tumour cell's ability to repair radiation-induced DNA double-strand breaks. When radiation creates breaks and cisplatin simultaneously prevents repair, the combined cell kill is synergistic — greater than either modality alone.
Side effects of cisplatin [2]:
| Side Effect | Mechanism | Management |
|---|---|---|
| Nephrotoxicity [2] | Cisplatin concentrates in renal tubular cells → direct tubular cell damage → acute tubular necrosis. Also causes renal magnesium wasting | Aggressive IV hydration before and after cisplatin; monitor creatinine; dose-reduce or switch to carboplatin if significant renal impairment |
| Ototoxicity [2] | Damages outer hair cells of the cochlea (particularly high-frequency hearing) → irreversible sensorineural hearing loss | Baseline and serial audiograms. High-frequency hearing loss is cumulative and dose-dependent. No effective prevention; dose modification if severe |
| Nausea and vomiting (76–100%) [2] | Cisplatin is the most emetogenic chemotherapy agent. Stimulates 5-HT3 receptors in the chemoreceptor trigger zone and GI tract | Triple antiemetic prophylaxis: 5-HT3 antagonist (ondansetron) + NK1 antagonist (aprepitant) + dexamethasone |
| Myelosuppression | Bone marrow suppression → neutropenia, anaemia, thrombocytopenia | Monitor FBC; G-CSF if needed; dose modification |
| Electrolyte disturbance | Renal magnesium and potassium wasting | Monitor and replace Mg²⁺ and K⁺ |
- Antimetabolite [2] — "anti" = against, "metabolite" = normal biochemical; 5-FU mimics uracil (a pyrimidine base) and tricks the cell's synthetic machinery
- Pyrimidine analog which interferes with DNA and RNA synthesis [2]
- F-dUMP inhibits thymidylate synthetase and thus depletes thymidine triphosphate which is a necessary component of DNA synthesis [2] — without thymidine, the cell cannot make new DNA → S-phase arrest → cell death
- F-UMP incorporates into RNA to replace uracil and inhibits cell growth [2] — disrupts RNA processing and function
Side effects of 5-FU [2]:
| Side Effect | Mechanism |
|---|---|
| Myelosuppression (pancytopenia) [2] | Bone marrow cells are rapidly dividing → susceptible to antimetabolite effects |
| Alopecia [2] | Hair follicle matrix cells are rapidly dividing → damaged by 5-FU |
| Stomatitis [2] | Oral mucosal cells turn over rapidly → ulceration |
| Diarrhoea [2] | GI mucosal cells turn over rapidly → mucosal damage → secretory diarrhoea |
| Hand-foot syndrome | 5-FU accumulates in palms and soles → painful erythema and desquamation |
| DPD deficiency risk | Dihydropyrimidine dehydrogenase (DPD) metabolizes 5-FU. Patients with DPD deficiency have catastrophic toxicity → can be fatal. DPD testing before 5-FU is now recommended |
- Nucleoside analogue (deoxycytidine analogue) that inhibits DNA synthesis
- Used in the GP (Gemcitabine + Cisplatin) induction regimen — now a preferred induction regimen for locoregionally advanced NPC based on the Sun Yat-Sen University trial (Zhang et al., NEJM 2019)
- Side effects: myelosuppression, flu-like symptoms, transaminase elevation
This is a newer but increasingly important modality for NPC, particularly recurrent/metastatic disease.
- Rationale: NPC (especially non-keratinizing undifferentiated) has a dense lymphocytic infiltrate and high PD-L1 expression. Tumour cells upregulate PD-L1 to evade immune surveillance → anti-PD-1 antibodies block this interaction → reactivate cytotoxic T cells against tumour
- Agents: Pembrolizumab, nivolumab, camrelizumab, toripalimab, tislelizumab
- Current indications:
- Recurrent/metastatic NPC after failure of platinum-based chemotherapy — anti-PD-1 monotherapy shows response rates of ~20–30%
- First-line metastatic NPC — GP + anti-PD-1 (e.g., JUPITER-02 trial: camrelizumab + GP vs GP alone showed improved PFS and OS) is now standard of care for first-line treatment of recurrent/metastatic NPC in many centres
- Combined with CRT for locoregionally advanced NPC — multiple ongoing trials (e.g., RATIONALE-309 with tislelizumab); early results are promising
- Side effects: Immune-related adverse events (irAEs) — pneumonitis, hepatitis, colitis, thyroiditis, dermatitis, hypophysitis. These are autoimmune-mediated because checkpoint blockade releases the brakes on the entire immune system, not just anti-tumour immunity
4. Surgical Treatment
General features [2]:
| Indication | Procedure | Details |
|---|---|---|
| Small local recurrence [2] | Nasopharyngectomy [2] | For patients with isolated nasopharyngeal recurrence after primary RT/CRT who are not suitable for re-irradiation or have already been re-irradiated. Endoscopic nasopharyngectomy (endoscopic endonasal approach) is increasingly used for selected cases. Open nasopharyngectomy (e.g., maxillary swing approach) for larger recurrences |
| Residual nodal disease following radiotherapy [2] | Neck dissection [2] | Neck dissection may be indicated in patients with residual nodal disease following radiotherapy or an isolated neck recurrence [2]. This is typically a planned neck dissection 8–12 weeks after CRT if PET-CT shows persistent FDG-avid residual nodes, or an unplanned salvage neck dissection for isolated nodal recurrence during follow-up |
| Contraindication | Explanation |
|---|---|
| Standard primary NPC (any stage) | RT/CRT is the standard of care; surgery is inferior and more morbid |
| Bulky or infiltrative recurrence | Unresectable due to skull base, carotid, or intracranial involvement |
| Unfit patient | Poor performance status, significant comorbidities precluding general anaesthesia |
5. Post-Treatment Follow-Up
Post-treatment follow-up [2]:
- Post-treatment baseline MRI of skull base and neck [2] — establishes the new post-treatment baseline for comparison on future surveillance imaging
- PET-CT scan 3 months after treatment completion [2] — assesses metabolic response; timing is important (waiting 3 months reduces false positives from post-radiation inflammation)
- Post-treatment EBV DNA levels [2]
- Post-radiotherapy plasma EBV DNA is validated as the MOST significant prognostic biomarker to select high-risk patients for adjuvant chemotherapy [2]
- Interpretation:
- Undetectable post-treatment → excellent prognosis, continue routine surveillance
- Detectable/persistent post-treatment → high risk of relapse → consider adjuvant chemotherapy and/or intensified surveillance
- Rising during follow-up → likely recurrence → PET-CT + MRI to localise; consider re-biopsy
| Time Post-Treatment | Investigations |
|---|---|
| 3 months | Post-treatment MRI + PET-CT + plasma EBV DNA |
| Every 3–6 months (years 1–2) | Clinical examination, nasopharyngoscopy, plasma EBV DNA |
| Every 6–12 months (years 3–5) | Clinical examination, nasopharyngoscopy, plasma EBV DNA, annual MRI |
| Annually (beyond 5 years) | Late effects monitoring (thyroid function, hearing, dental, temporal lobes) |
| Scenario | Treatment Options |
|---|---|
| Isolated local recurrence (nasopharynx) | Re-irradiation (IMRT, stereotactic body RT, brachytherapy) OR nasopharyngectomy (endoscopic or open). Re-irradiation carries significant risk of late toxicity (temporal lobe necrosis, cranial neuropathy, carotid blowout) |
| Isolated regional recurrence (neck) | Salvage neck dissection [2] ± re-irradiation |
| Distant metastasis (first-line) | GP + anti-PD-1 (gemcitabine + cisplatin + camrelizumab/pembrolizumab) — current standard of care per JUPITER-02 and CAPTAIN-1st trials |
| Distant metastasis (second-line) | Anti-PD-1 monotherapy; or other chemotherapy doublets (capecitabine + cisplatin); clinical trials |
| Oligometastatic disease | Consider local treatment to metastatic sites (surgery, SBRT) + systemic therapy — selected patients may achieve long-term disease control |
Management framework general principles [16][18]:
- Tumour clearance with long-term survival benefit [18]
- Organ and function preservation [18]
- When surgery is indicated → resection with adequate margins, reconstruction for form and function [18]
- Rehabilitation always — swallowing, voice, and hearing [18]
For NPC specifically:
- Surgery is almost never first-line
- RT is the backbone; chemotherapy (cisplatin) is added as a radiosensitizer for Stage II+
- Immunotherapy is transforming the metastatic/recurrent setting
- Plasma EBV DNA guides adjuvant therapy decisions and surveillance
- Late effects monitoring is critical (thyroid, hearing, dental, temporal lobes, cranial nerves)
High Yield Summary — Management of NPC
Key exception in H&N cancer: NPC uses chemo-irradiation in late stage (NOT surgery). Oral cavity and thyroid use surgery in early stage.
Stage-based approach:
- Stage I → RT alone (> 90% cure rate)
- Stage II–IVB → Concurrent CRT (cisplatin + IMRT) ± induction GP ± adjuvant chemo
- Stage IVC → Palliative chemotherapy ± immunotherapy ± palliative RT
RT: IMRT is standard. Bilateral neck irradiation always (bilateral nodal drainage). Side effects: mucositis, xerostomia, ototoxicity, temporal lobe necrosis.
Chemotherapy: Cisplatin (DNA cross-linker; nephrotoxic, ototoxic, highly emetogenic). 5-FU (thymidylate synthetase inhibitor; mucositis, myelosuppression). Gemcitabine + Cisplatin = preferred induction regimen.
Immunotherapy: Anti-PD-1 (camrelizumab, pembrolizumab) — now standard first-line for metastatic NPC combined with GP.
Surgery: NOT first-line. Reserved for salvage: nasopharyngectomy for small local recurrence, neck dissection for residual/recurrent nodal disease.
Post-treatment: MRI + PET-CT at 3 months. Plasma EBV DNA is the MOST significant prognostic biomarker for selecting adjuvant therapy and detecting recurrence.
Active Recall - Management of Nasopharyngeal Carcinoma
References
[2] Senior notes: felixlai.md (felix:364, 365, 366) — NPC diagnosis, treatment, screening [16] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p43) — Management framework [18] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p44) — Management framework general principles
Complications of Nasopharyngeal Carcinoma
Complications of NPC can be organized into three broad categories: (A) complications of the disease itself (from tumour progression and metastasis), (B) complications of treatment (radiotherapy, chemotherapy, immunotherapy, surgery), and (C) long-term survivorship issues. Because NPC is treated primarily with radiation-based therapy (not surgery), the treatment complication profile is dominated by radiation-related toxicity — and given that many NPC patients are diagnosed young (40–60 years) and achieve long survival, late effects are a major clinical concern.
Let me walk through each systematically, always connecting back to the mechanism.
A. Complications of the Disease (Tumour-Related)
These arise from local invasion, regional spread, and distant metastasis — essentially what happens if the tumour is left untreated or progresses despite treatment.
| Complication | Mechanism |
|---|---|
| Cranial nerve palsies (CN III–VI, IX–XII) | Superior spread through foramen lacerum → cavernous sinus (CN III, IV, V1, V2, VI). Lateral spread through parapharyngeal space → CN IX, X, XI, XII. The nasopharynx sits at the crossroads of these neurovascular structures [2]. CN VI (abducens) is most commonly affected because it has the longest intracranial course and traverses the cavernous sinus |
| Cavernous sinus syndrome | Tumour invades the cavernous sinus via foramen lacerum → compression of CN III, IV, V1, V2, VI + internal carotid artery (ICA). Presents as complete ophthalmoplegia, ptosis, periorbital numbness, and potentially Horner syndrome (involvement of sympathetic plexus around ICA) |
| Eustachian tube obstruction → serous otitis media → conductive hearing loss | Tumour at the fossa of Rosenmüller compresses or infiltrates the Eustachian tube opening and the tensor/levator veli palatini muscles → failure to equalize middle ear pressure → fluid transudation into the middle ear. Chronic effusion can lead to tympanic membrane retraction and cholesteatoma |
| Nasal obstruction and epistaxis | Anterior extension into the nasal cavity → mechanical obstruction of the posterior choana; friable tumour vasculature bleeds easily |
| Skull base erosion | Superior extension destroys bone of the clivus, sphenoid sinus floor, or petrous apex. Loss of the bony barrier allows intracranial extension → risk of meningitis, CSF leak (rare), brain compression |
| Carotid artery encasement | Lateral extension into the parapharyngeal space can encase or erode the internal carotid artery → risk of carotid blowout syndrome (catastrophic haemorrhage) — though this is more commonly a post-treatment complication |
| Dysphagia and aspiration | CN IX and X palsy (from parapharyngeal invasion) → pharyngeal motor dysfunction → inability to protect the airway during swallowing → aspiration pneumonia |
| Complication | Mechanism |
|---|---|
| Airway compromise | Massive bilateral cervical lymphadenopathy can compress the airway externally, though this is rare. Retropharyngeal nodal disease can cause posterior pharyngeal wall bulging → narrowing of the airway. Always protect the airway for all H&N cancer [3] |
| Jugular vein compression/thrombosis | Large level II nodes can compress or invade the internal jugular vein → venous congestion, facial oedema |
| Brachial plexopathy | N3 nodes extending below the cricoid (supraclavicular fossa) can involve the brachial plexus → upper limb weakness, pain, paraesthesia |
Common sites of distant metastasis: Bone (75%) / Liver / Lung / Distant lymph nodes [2]:
| Complication | Mechanism |
|---|---|
| Pathological fractures | Bone metastasis (most common, ~75%) [2] — tumour in bone marrow stimulates osteoclast activity → bone destruction → structural weakness → fracture with minimal or no trauma. Commonly affects the spine (compression fractures → spinal cord compression), pelvis, ribs, long bones |
| Spinal cord compression | Vertebral body collapse or epidural tumour extension → compression of the spinal cord → neurological emergency. Presents with back pain, lower limb weakness, sensory level, bladder dysfunction. Requires urgent MRI and dexamethasone + RT/surgery |
| Hepatic failure | Extensive liver metastasis → replacement of functional hepatocytes → liver failure (jaundice, coagulopathy, encephalopathy). Usually signifies end-stage disease |
| Respiratory compromise | Pulmonary metastases → reduced gas exchange area → dyspnoea. Lymphangitis carcinomatosis → diffuse interstitial tumour spread in pulmonary lymphatics → severe dyspnoea |
| Hypercalcaemia of malignancy | Bone metastasis + PTHrP secretion → ↑ serum calcium → confusion, polyuria, constipation, cardiac arrhythmias. Medical emergency requiring IV normal saline, bisphosphonates (zoledronic acid), and calcitonin |
| Bone marrow failure | Extensive marrow infiltration → pancytopenia → infection risk, anaemia, bleeding |
| Complication | Mechanism |
|---|---|
| Dermatomyositis [2] | NPC is one of the malignancies specifically associated with paraneoplastic dermatomyositis, particularly in Chinese populations. Autoimmune-mediated inflammatory myopathy triggered by tumour antigens that cross-react with muscle proteins. Presents with proximal muscle weakness, heliotrope rash (periorbital violet discolouration), Gottron papules (erythematous plaques over knuckles). May precede, coincide with, or follow NPC diagnosis. Treatment of the underlying NPC can improve the dermatomyositis |
B. Complications of Treatment
This is the high-yield section because NPC patients often survive long-term, and treatment toxicity significantly impacts quality of life.
1. Radiotherapy Complications
Radiotherapy is the backbone of NPC treatment. The nasopharynx's anatomical position means that the radiation field inevitably exposes multiple critical surrounding structures. Complications are classified by timing:
| Complication | Mechanism | Pathophysiological Basis |
|---|---|---|
| Mucositis (mouth and throat sores) [2] | Radiation damages rapidly dividing mucosal epithelial cells of the oropharynx, oral cavity, and nasopharynx → mucosal breakdown → ulceration → pain, dysphagia, secondary infection (candidiasis, bacterial) | Mucosal epithelium has a turnover of ~5–7 days → highly radiosensitive. Damage outpaces regeneration during the treatment course |
| Dermatitis | Radiation damages skin in the treatment field → erythema → dry desquamation → moist desquamation (severe) | Similar to sunburn physiology — radiation-induced free radical damage to basal keratinocytes |
| Xerostomia (dry mouth) — begins acutely, becomes chronic | Radiation damages salivary gland acinar cells. Serous acini (parotid gland) are MORE radiosensitive than mucous acini (submandibular/sublingual). Loss of serous saliva → thick, sticky, reduced-volume saliva | The parotid gland is the largest serous salivary gland and lies within or adjacent to the radiation field. IMRT has reduced parotid damage by sparing the contralateral parotid, but some degree of xerostomia is almost universal |
| Dysgeusia (taste disturbance) | Radiation damages taste buds (especially on the posterior tongue and soft palate, which are in the field) and reduces salivary flow (saliva is needed to dissolve tastants and deliver them to taste receptors) | Usually partially reversible over months; some permanent loss is common |
| Odynophagia / Dysphagia | Pharyngeal mucosal inflammation → oedema → pain on swallowing → difficulty maintaining nutrition. May require nasogastric tube (NGT) or percutaneous endoscopic gastrostomy (PEG) during treatment | Weight loss during CRT is a major clinical problem; nutritional support is critical |
| Myelosuppression (when concurrent chemotherapy given) | Cisplatin + radiation synergistically suppress bone marrow → neutropenia (infection risk), thrombocytopenia (bleeding), anaemia (fatigue) | Monitor FBC weekly during CRT; hold chemotherapy if severe neutropenia |
| Complication | Mechanism |
|---|---|
| Lhermitte sign | Transient demyelination of the posterior columns of the spinal cord (within the radiation field) → electric shock-like sensation radiating down the spine on neck flexion. Self-limiting (resolves over months). NOT a sign of myelopathy |
| Persistent xerostomia | Ongoing salivary gland damage. Parotid function may partially recover over 12–24 months with IMRT |
These are the most important complications from an exam and clinical perspective because they are irreversible, cumulative, and affect survivors for decades.
| Complication | Mechanism | Clinical Significance |
|---|---|---|
| Temporal lobe necrosis (TLN) | The temporal lobes sit directly above and lateral to the nasopharynx → inevitably receive significant radiation dose. High-dose radiation → white matter vasculopathy (endothelial damage → thrombosis of small vessels → ischaemic necrosis of brain parenchyma). Presents with headache, cognitive decline, personality change, seizures, focal neurological deficits. Diagnosed on MRI (ring-enhancing lesion in temporal lobe, often with surrounding oedema — can mimic brain metastasis or abscess). IMRT has reduced but not eliminated this risk | One of the most devastating late effects. Incidence: ~5% at 10 years with IMRT. Treatment: dexamethasone, bevacizumab (anti-VEGF, reduces peritumoural oedema), hyperbaric oxygen, surgical resection for refractory cases |
| Sensorineural hearing loss (SNHL) | Radiation damages cochlear hair cells (outer hair cells are most vulnerable) and cochlear vasculature → irreversible high-frequency hearing loss. Additive with cisplatin ototoxicity — both mechanisms target the cochlea | Cisplatin causes ototoxicity [2]. Radiation compounds this. Patients may have BOTH conductive loss (from Eustachian tube dysfunction/middle ear effusion) and sensorineural loss (from cochlear damage). Serial audiograms needed. Hearing aids for rehabilitation |
| Osteoradionecrosis (ORN) of the mandible | Radiation damages osteocytes and periosteal blood supply → hypoxic, hypovascular, hypocellular bone → failure of wound healing → avascular necrosis. Triggered by dental extraction or trauma post-RT (irradiated bone cannot mount an adequate healing response) | Pre-RT dental assessment and extraction of hopeless teeth is mandatory. Post-RT extractions carry 5–15% risk of ORN. Management: conservative (antibiotics, HBO), surgical (sequestrectomy, free flap reconstruction for severe cases) |
| Trismus (restricted mouth opening) | Radiation fibrosis of masticator muscles (medial/lateral pterygoids, masseter, temporalis) and temporomandibular joint (TMJ) capsule → progressive loss of jaw mobility | Incidence: 25–40%. Preventable with jaw stretching exercises (TheraBite) started during RT and continued indefinitely. Severe trismus ( < 20 mm interincisal distance) significantly impairs eating, dental care, and quality of life |
| Radiation-induced cranial neuropathy | Radiation damages the vasa nervorum of cranial nerves within the treatment field → ischaemic neuropathy → delayed CN palsy (most commonly CN IX, X, XI, XII — lower cranial nerves). Presents months to years post-RT | Can cause dysphagia (CN IX, X), aspiration, vocal cord palsy (hoarseness), tongue weakness (CN XII). Distinguished from tumour recurrence by stable imaging and undetectable plasma EBV DNA |
| Dysphagia / Pharyngeal stenosis | Radiation fibrosis of pharyngeal constrictor muscles → loss of pharyngeal peristalsis → difficulty swallowing. Progressive stricture formation → narrowing of the pharyngeal lumen | Rehabilitation always — swallowing, voice, and hearing [18]. Swallowing exercises must start during RT and continue long-term. PEG dependence rate post-CRT is ~5–10% |
| Hypothyroidism | Radiation to the neck fields damages thyroid follicular cells → gradual loss of thyroid hormone production → primary hypothyroidism (↑ TSH, ↓ fT4) | Incidence: 20–40% at 5 years. Requires lifelong thyroid function monitoring and levothyroxine replacement |
| Carotid artery stenosis / Stroke | Radiation accelerates atherosclerosis in the carotid arteries (radiation-induced endothelial injury → intimal hyperplasia → accelerated plaque formation) → carotid stenosis → increased stroke risk | Onset typically > 5 years post-RT. Annual carotid duplex ultrasound surveillance recommended. Antiplatelet therapy and carotid intervention (endarterectomy/stenting) if significant stenosis |
| Carotid blowout syndrome (CBS) | Radiation weakens the arterial wall (adventitial damage, vasa vasorum obliteration) → pseudoaneurysm formation → rupture → catastrophic haemorrhage. Risk increases with re-irradiation, local recurrence, and wound breakdown | Life-threatening emergency. Presents with sentinel bleed (small warning bleed from ear, nose, or mouth) followed by massive haemorrhage. Immediate management: direct pressure, airway protection, emergency angiography with embolisation or stent placement |
| Nasopharyngeal necrosis / Skull base osteonecrosis | High-dose radiation to the nasopharynx → mucosal and bone necrosis → crusting, foul-smelling discharge, pain. Can mimic local recurrence on imaging | Distinguished from recurrence by biopsy (necrotic tissue without viable tumour) and undetectable plasma EBV DNA |
| Secondary malignancy | Radiation-induced DNA damage in normal tissues within the field → second primary cancer years to decades later (e.g., sarcoma of irradiated field, thyroid cancer, brain tumour) | Risk increases with time. Overall incidence ~1–3% at 20 years. Highlights the importance of long-term surveillance |
The Big Five Late Effects of NPC Radiotherapy
For exams, remember the five most important late effects — use the mnemonic "TOXIN":
- T = Temporal lobe necrosis
- O = Osteoradionecrosis (mandible)
- X = Xerostomia (chronic)
- I = (H)Ipothyroidism (hypothyroidism)
- N = Neuropathy (cranial nerve, sensorineural hearing loss)
These were covered in the Management section but are summarised here for completeness:
| Agent | Key Complications | Mechanism |
|---|---|---|
| Cisplatin [2] | Nephrotoxicity | Direct tubular cell toxicity; renal Mg²⁺ wasting |
| Ototoxicity | Outer hair cell damage in cochlea → irreversible high-frequency SNHL; cumulative and dose-dependent | |
| Nausea and vomiting (76–100%) | Most emetogenic chemotherapy agent; 5-HT3 receptor stimulation in chemoreceptor trigger zone | |
| Myelosuppression | Bone marrow suppression → neutropenia, anaemia, thrombocytopenia | |
| Electrolyte disturbance | Hypomagnesaemia, hypokalaemia from renal wasting | |
| Peripheral neuropathy | Sensory neuropathy (glove-and-stocking distribution) from axonal damage; dose-limiting | |
| 5-Fluorouracil [2] | Myelosuppression (pancytopenia) | Bone marrow cells rapidly dividing → susceptible to antimetabolite effect |
| Stomatitis | Oral mucosal cells rapidly dividing → ulceration | |
| Diarrhoea | GI mucosal cells rapidly dividing → mucosal damage | |
| Alopecia | Hair follicle matrix cells rapidly dividing → hair loss | |
| DPD deficiency catastrophe | Patients lacking dihydropyrimidine dehydrogenase cannot metabolize 5-FU → severe/fatal toxicity | |
| Gemcitabine | Myelosuppression, hepatotoxicity | Nucleoside analogue disrupts DNA synthesis in rapidly dividing cells |
Anti-PD-1 agents (pembrolizumab, camrelizumab, toripalimab) release the immune "brakes" globally, not just against the tumour:
| Organ System | irAE | Mechanism |
|---|---|---|
| Lung | Immune pneumonitis | T-cell infiltration of lung parenchyma → inflammatory alveolitis → cough, dyspnoea, ground-glass opacities on CT |
| Liver | Immune hepatitis | T-cell mediated hepatocyte destruction → ↑ AST/ALT |
| Colon | Immune colitis | T-cell infiltration of colonic mucosa → diarrhoea, abdominal pain (mimics IBD) |
| Thyroid | Thyroiditis | Destructive thyroiditis → transient thyrotoxicosis → subsequent hypothyroidism (on top of radiation-induced hypothyroidism) |
| Skin | Dermatitis, vitiligo | T-cell mediated skin inflammation; vitiligo can actually be a marker of good anti-tumour response |
| Endocrine | Hypophysitis, adrenalitis | T-cell infiltration of pituitary or adrenal glands → hypopituitarism, adrenal insufficiency |
| Cardiac | Myocarditis | Rare but potentially fatal; T-cell infiltration of myocardium → fulminant heart failure |
Management of irAEs: Hold immunotherapy, systemic corticosteroids (prednisolone/methylprednisolone), and for refractory cases, additional immunosuppression (infliximab, mycophenolate).
Since surgery is only used in the salvage setting (post-RT tissue), complications are significantly higher than in primary surgery:
| Complication | Mechanism |
|---|---|
| Carotid blowout | Post-RT carotid wall is weakened; surgical dissection near the carotid in irradiated tissue further compromises the wall → catastrophic haemorrhage |
| Wound breakdown / Fistula | Irradiated tissues have poor vascularity and healing capacity → wound dehiscence, pharyngocutaneous fistula |
| Cranial nerve injury | Salvage nasopharyngectomy near the skull base risks injury to CN V, VI, internal carotid, CN IX–XII. Post-RT fibrosis obliterates normal tissue planes → higher injury risk |
| CSF leak | Skull base surgery or re-irradiation can breach the dura → CSF rhinorrhoea → risk of meningitis |
| Aspiration pneumonia | If CN IX/X already compromised by tumour or prior RT, salvage surgery further impairs pharyngeal function → aspiration |
General principles of H&N cancer management apply [18]:
- Tumour clearance with long-term survival benefit [18]
- Organ and function preservation [18]
- Rehabilitation always — swallowing, voice, and hearing [18]
NPC survivors face a unique constellation of long-term challenges:
| Domain | Issues | Management |
|---|---|---|
| Swallowing | Pharyngeal fibrosis, dysphagia, aspiration risk, PEG dependence | Swallowing exercises (pre-RT through long-term), speech-language pathology, modified diet consistency |
| Hearing | Combined conductive (middle ear effusion) + sensorineural (cochlear damage from RT + cisplatin) | Audiological assessment, hearing aids, consideration of myringotomy/grommets for persistent effusion |
| Speech | Velopharyngeal insufficiency (radiation damage to soft palate → nasal regurgitation, hypernasal speech), tongue weakness (CN XII neuropathy) | Speech therapy |
| Dental | Xerostomia → rampant dental caries (saliva normally buffers acid and remineralizes enamel; without it, teeth decay rapidly), ORN risk | Lifelong fluoride trays, saliva substitutes/sialogogues (pilocarpine), meticulous oral hygiene, avoid post-RT dental extractions if possible |
| Endocrine | Hypothyroidism, hypopituitarism (if pituitary in field) | Regular thyroid function tests, pituitary hormone panels if symptomatic |
| Neurocognitive | Temporal lobe necrosis → memory impairment, personality change, seizures | Neuropsychological assessment, dexamethasone, bevacizumab, antiepileptics |
| Psychological | Depression, anxiety, body image issues, fear of recurrence | Psycho-oncology support, patient support groups |
| Cardiovascular | Accelerated carotid atherosclerosis → stroke risk | Cardiovascular risk factor management, carotid surveillance |
The NPC Survivor Clinic Checklist
At every follow-up visit for an NPC survivor, systematically assess:
- Recurrence → plasma EBV DNA, clinical examination, nasopharyngoscopy
- Swallowing → can they eat? Aspiration risk?
- Hearing → audiometry
- Thyroid → TSH, fT4
- Dental → oral examination, fluoride compliance
- Neurological → cranial nerves, cognition, temporal lobe symptoms
- Vascular → carotid surveillance if > 5 years post-RT
- Psychological → mood screening
| Scenario | Complication | Management |
|---|---|---|
| Local recurrence | Re-treatment carries very high toxicity (temporal lobe necrosis, carotid blowout from re-irradiation). Nasopharyngeal necrosis. Skull base osteonecrosis | Surgery (open maxillary swing, endoscopic or robotic nasopharyngectomy), chemotherapy, second dose radiotherapy, immunotherapy [19] |
| Regional recurrence | Re-irradiation toxicity if already treated; surgical salvage in irradiated field has high wound complication rate | Neck dissection may be indicated for residual nodal disease following radiotherapy or isolated neck recurrence [2] |
| Distant relapse | Bone pain, pathological fractures, liver failure, respiratory failure — generally incurable, treatment is palliative | Systemic therapy (GP + anti-PD-1); palliative RT to symptomatic sites; best supportive care |
High Yield Summary — Complications of NPC
Disease complications: Cranial nerve palsies (CN VI most common), cavernous sinus syndrome, serous otitis media, bone metastasis (75%) with pathological fractures and spinal cord compression, dermatomyositis (paraneoplastic).
Treatment complications — RT (most important):
- Acute: mucositis, dermatitis, xerostomia, dysphagia
- Late (Big Five = TOXIN): Temporal lobe necrosis, Osteoradionecrosis of mandible, Xerostomia (chronic), HIpothyroidism, Neuropathy (cranial nerve, SNHL)
- Also: trismus, carotid stenosis/stroke, carotid blowout, pharyngeal stenosis, secondary malignancy
Treatment complications — Chemotherapy: Cisplatin = nephrotoxicity, ototoxicity, severe emesis. 5-FU = myelosuppression, mucositis, diarrhoea, DPD deficiency risk.
Treatment complications — Immunotherapy: irAEs (pneumonitis, hepatitis, colitis, thyroiditis, myocarditis).
Survivorship: Lifelong monitoring of hearing, thyroid, dental health, swallowing, cognition, carotid vasculature, and psychological well-being. Rehabilitation always — swallowing, voice, hearing.
Pre-RT dental assessment is mandatory — post-RT dental extraction risks osteoradionecrosis.
Active Recall - Complications of Nasopharyngeal Carcinoma
References
[2] Senior notes: felixlai.md (felix:357, 364, 365) — NPC overview, clinical features, treatment side effects [3] Senior notes: felixlai.md (felix:342) — H&N cancer overview, airway protection [18] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p44) — Management framework general principles: tumour clearance, organ preservation, rehabilitation [19] Lecture slides: GC 215. Common nasal conditions and nasopharyngeal carcinoma (1).pdf (p55) — NPC treatment: early stage IMRT, late stage concurrent CRT, residual/recurrence management
High Yield Summary
Definition: Malignant epithelial neoplasm of the nasopharynx, most commonly arising from the fossa of Rosenmüller.
Epidemiology: Endemic in Southern China/Hong Kong. 10th most common cancer in HK, 6th in males. M:F = 2.5:1. > 70% of global cases in East/Southeast Asia.
Etiology triad: (1) EBV (primary agent; detected in nearly 100% of non-keratinizing NPC; key markers: VCA-IgA, plasma EBV DNA), (2) Genetics (HLA haplotypes, CYP2A6, family history), (3) Environmental (salted fish/preserved foods with nitrosamines, smoking, alcohol).
Pathology: > 95% non-keratinizing in endemic areas (undifferentiated most common — best prognosis, most radiosensitive). Keratinizing = sporadic form. Basaloid = worst prognosis.
Key anatomy: Nasopharynx → fossa of Rosenmüller (commonest origin). Lateral spread → Eustachian tube (effusion), parapharyngeal space (CN IX-XII, Horner). Superior spread → skull base, cavernous sinus (CN III-VI). Rich lymphatic drainage → early bilateral LN metastasis.
Clinical features:
- Neck mass (most common presenting complaint) — bilateral cervical LN, especially level II
- Nasal: epistaxis, obstruction
- Otological: unilateral serous otitis media (critical clue in adults), conductive hearing loss
- Neurological: CN VI palsy (most common CN), CN V numbness, cavernous sinus syndrome
- Distant metastasis: bone (75%) > liver > lung
Red flag: Any adult with unilateral middle ear effusion in an NPC-endemic region → must scope the nasopharynx.
N staging is unique to NPC — based on laterality, size, and position relative to cricoid (not ENE like other H&N cancers).
High Yield Summary — Differential Diagnosis of NPC
Nasopharyngeal mass DDx: NPC (most common malignancy), NHL (especially NK/T-cell lymphoma in HK), JNA (adolescent males — DO NOT biopsy), sinonasal carcinoma extending posteriorly, minor salivary gland tumour, melanoma, rhabdomyosarcoma (children).
Neck mass DDx: Metastatic NPC, other metastatic H&N SCC (oropharyngeal HPV+, hypopharyngeal, laryngeal), lymphoma (Hodgkin/NHL), thyroid carcinoma, TB lymphadenitis, branchial cleft cyst (beware in adults > 40 — likely cystic met), paraganglioma, schwannoma.
Key differentiating tools: FNA with EBV PCR (NPC) vs HPV PCR (oropharyngeal SCC); EBER-ISH on biopsy; plasma EBV DNA; p16 IHC; imaging characteristics (JNA = intensely vascular, do not biopsy).
Search for occult primary: Nasopharyngoscopy + bilateral tonsillectomy + tongue base/hypopharynx biopsy + panendoscopy. FNA of node with EBV/HPV PCR guides the search.
Red flag: Any unilateral nasal symptom or adult unilateral middle ear effusion → exclude nasopharyngeal neoplasm.
High Yield Summary — Diagnosis of NPC
Definitive diagnosis: Endoscope-guided biopsy of the nasopharyngeal primary → histopathology (WHO classification) + EBER-ISH (EBV confirmation).
Do NOT excise/biopsy neck nodes — NPC is treated with RT/CRT, not surgery. Nodal surgery compromises treatment.
Key biomarkers:
- Plasma EBV DNA (quantitative PCR) — diagnosis, staging, prognosis, treatment monitoring, recurrence detection. The single most important biomarker.
- EBV VCA-IgA and EA-IgA — screening use but low specificity. Titre may precede diagnosis by up to 10 years.
- ALP — elevated suggests bone metastasis.
FNA of neck node: Cytology + EBV PCR (for NPC) / HPV PCR (for oropharyngeal SCC). Guides search for primary. Does NOT provide tissue architecture.
Imaging:
- MRI = primary modality for T and N staging (superior soft tissue contrast).
- PET-CT = primary modality for M staging (superior for bone and distant metastases).
- CT = complementary for bone erosion; CT chest/abdomen if PET-CT unavailable.
- Bone scan / CXR = adjuncts if PET-CT unavailable.
Panendoscopy = exclude synchronous primary (10% risk from field cancerization).
Screening: Plasma EBV DNA (two-time-point approach) in high-risk populations → nasopharyngoscopy + MRI if persistently positive.
High Yield Summary — Management of NPC
Key exception in H&N cancer: NPC uses chemo-irradiation in late stage (NOT surgery). Oral cavity and thyroid use surgery in early stage.
Stage-based approach:
- Stage I → RT alone (> 90% cure rate)
- Stage II–IVB → Concurrent CRT (cisplatin + IMRT) ± induction GP ± adjuvant chemo
- Stage IVC → Palliative chemotherapy ± immunotherapy ± palliative RT
RT: IMRT is standard. Bilateral neck irradiation always (bilateral nodal drainage). Side effects: mucositis, xerostomia, ototoxicity, temporal lobe necrosis.
Chemotherapy: Cisplatin (DNA cross-linker; nephrotoxic, ototoxic, highly emetogenic). 5-FU (thymidylate synthetase inhibitor; mucositis, myelosuppression). Gemcitabine + Cisplatin = preferred induction regimen.
Immunotherapy: Anti-PD-1 (camrelizumab, pembrolizumab) — now standard first-line for metastatic NPC combined with GP.
Surgery: NOT first-line. Reserved for salvage: nasopharyngectomy for small local recurrence, neck dissection for residual/recurrent nodal disease.
Post-treatment: MRI + PET-CT at 3 months. Plasma EBV DNA is the MOST significant prognostic biomarker for selecting adjuvant therapy and detecting recurrence.
High Yield Summary — Complications of NPC
Disease complications: Cranial nerve palsies (CN VI most common), cavernous sinus syndrome, serous otitis media, bone metastasis (75%) with pathological fractures and spinal cord compression, dermatomyositis (paraneoplastic).
Treatment complications — RT (most important):
- Acute: mucositis, dermatitis, xerostomia, dysphagia
- Late (Big Five = TOXIN): Temporal lobe necrosis, Osteoradionecrosis of mandible, Xerostomia (chronic), HIpothyroidism, Neuropathy (cranial nerve, SNHL)
- Also: trismus, carotid stenosis/stroke, carotid blowout, pharyngeal stenosis, secondary malignancy
Treatment complications — Chemotherapy: Cisplatin = nephrotoxicity, ototoxicity, severe emesis. 5-FU = myelosuppression, mucositis, diarrhoea, DPD deficiency risk.
Treatment complications — Immunotherapy: irAEs (pneumonitis, hepatitis, colitis, thyroiditis, myocarditis).
Survivorship: Lifelong monitoring of hearing, thyroid, dental health, swallowing, cognition, carotid vasculature, and psychological well-being. Rehabilitation always — swallowing, voice, hearing.
Pre-RT dental assessment is mandatory — post-RT dental extraction risks osteoradionecrosis.
Laryngeal Carcinoma
Laryngeal carcinoma is a malignant neoplasm arising from the epithelial lining of the larynx, most commonly squamous cell carcinoma, strongly associated with smoking and alcohol use, presenting with hoarseness, dysphagia, or stridor.
Neck Mass
A neck mass is a palpable swelling in the neck that may arise from congenital, inflammatory/infectious, or neoplastic causes involving lymph nodes, thyroid, salivary glands, or other cervical structures.