Head And Neck Cancer
Head and neck cancer refers to a group of malignancies arising from the squamous epithelial lining of the mucosal surfaces of the oral cavity, pharynx, larynx, nasal cavity, and paranasal sinuses, often associated with tobacco, alcohol use, and HPV infection.
Head and Neck Cancer
Head and neck (H&N) cancer refers to a heterogeneous group of malignancies arising from the mucosal surfaces of the upper aerodigestive tract (UADT). The term "head and neck" is a shorthand — break it down and it encompasses everything from the lips to the larynx, but excludes brain, eye, thyroid (usually discussed separately), and skin cancers (though skin SCC of the face can overlap).
- Most H&N cancers begin in the mucosal surfaces of the upper aerodigestive tract and are predominantly squamous cell carcinoma (SCC) [1][2]
- 90% of head and neck malignancies are squamous cell carcinoma (SCC) (not including nasopharynx and thyroid) [3]
- They arise from 5 anatomical areas: oral cavity, pharynx (nasopharynx, oropharynx, hypopharynx), larynx, nasal cavity & paranasal sinuses, and salivary glands [2]
The word "squamous" comes from Latin squama = scale — these are cancers of the flat, scale-like epithelial cells that line these mucosal surfaces. When you hear "HNSCC" = Head and Neck Squamous Cell Carcinoma, that is the dominant entity.
Airway First — Always
ALWAYS protect the airway for all H&N cancer [2]. This is the cardinal rule. H&N tumours can obstruct the airway acutely (tumour bulk, bleeding, post-operative oedema). Before you think about staging or treatment, secure the airway.
2. Epidemiology
- Incidence: H&N cancers (excluding nasopharynx) represent approximately 4–5% of all cancers globally, with an estimated 900,000+ new cases annually worldwide [4].
- Male preponderance — reflects the higher prevalence of smoking and alcohol use in males historically [2].
- Predominantly a disease of the elderly (age > 60) [2] — cumulative carcinogen exposure over decades.
- Exception — HPV-related oropharyngeal cancer: This is rising in younger populations (40s–50s), particularly in Western countries. These patients tend to be younger, non-smoking males with a history of oral sexual contact [2].
| Cancer Type | HK Relevance |
|---|---|
| Nasopharyngeal carcinoma (NPC) | Endemic in Southern China including Hong Kong [2]. 10th most common cancer overall; 6th in males. Incidence ~6–10/100,000 in HK (vs < 1/100,000 in Western countries). |
| Oral cavity SCC | Associated with smoking, alcohol, betel nut chewing (less common in HK than Southeast Asia but still relevant). |
| Oropharyngeal SCC | HPV-driven oropharyngeal cancer is increasing in HK, though still less common than in Western populations. |
| Laryngeal SCC | Strongly linked to smoking and alcohol; male predominance. |
High Yield: NPC in Hong Kong
NPC is the signature H&N cancer of Hong Kong. If you see a Southern Chinese patient with unilateral serous otitis media, epistaxis, or a neck lump — think NPC until proven otherwise.
- HPV-positive oropharyngeal cancer is increasing in incidence globally (especially in developed nations) while smoking-related H&N cancers are declining in many Western countries.
- NPC incidence in HK has been gradually declining over the past two decades, possibly related to changes in dietary habits (less salted fish consumption) and EBV screening programmes.
3. Anatomy and Function
Understanding H&N cancer requires knowing the anatomy cold. The UADT is divided into 5 basic anatomical areas, and each area has distinct clinical behaviour, lymphatic drainage, and treatment implications.
Boundaries: From the vermilion border of the lips anteriorly to the junction of the hard and soft palate superiorly and the circumvallate papillae of the tongue inferiorly.
Subsites:
- Lips (upper, lower, commissure)
- Anterior two-thirds of tongue (oral tongue)
- Floor of mouth
- Buccal mucosa
- Hard palate
- Alveolar ridges (upper and lower gingiva)
- Retromolar trigone (the area behind the last molar tooth)
Lymphatic drainage: Primarily to Level I (submental and submandibular nodes), then Level II and III. The tongue has notoriously rich lymphatic drainage and can skip-metastasize to Level III–IV.
Key functions: Mastication, articulation of speech, taste (anterior 2/3 tongue via CN VII chorda tympani), initiation of swallowing.
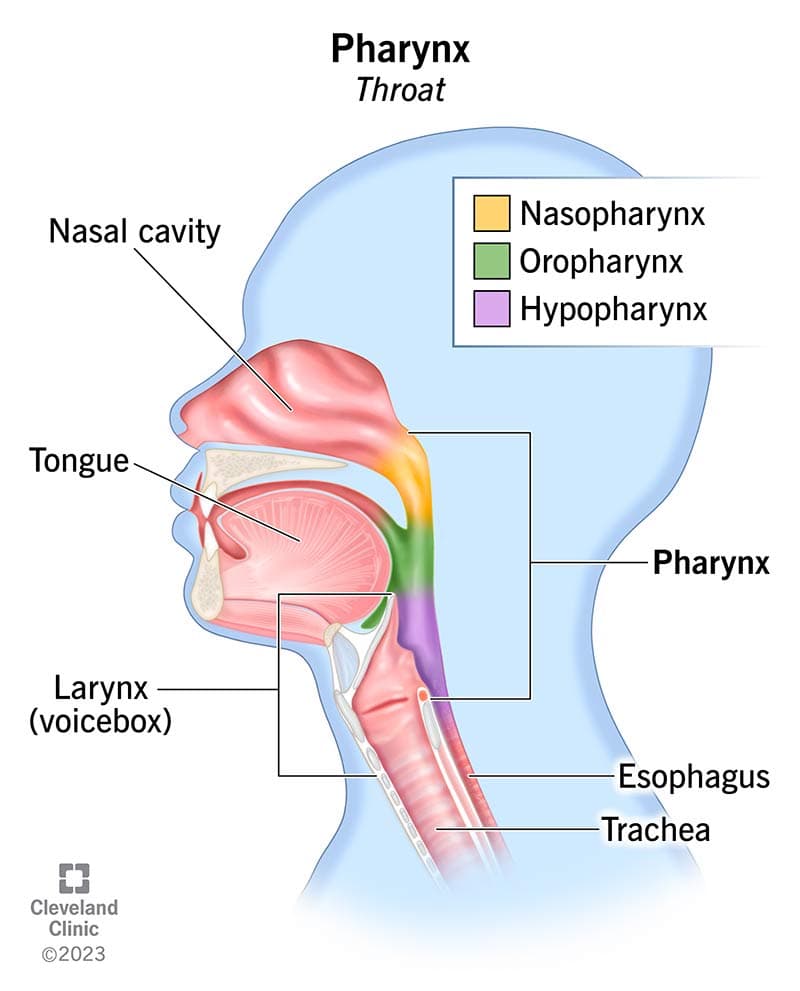
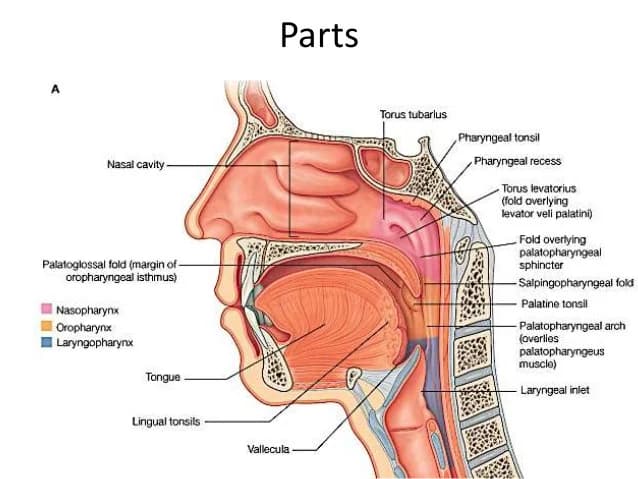
3.3 Pharynx
The pharynx is a muscular tube divided into three subsections from superior to inferior:
- Boundaries: From the skull base superiorly to the level of the soft palate inferiorly.
- Key landmark: Pharyngeal recess (Fossa of Rosenmüller) — the most common site of origin for NPC [2]. This is a mucosal recess lateral to the torus tubarius. It is "clinically occult" — meaning tumours here can grow silently for a long time.
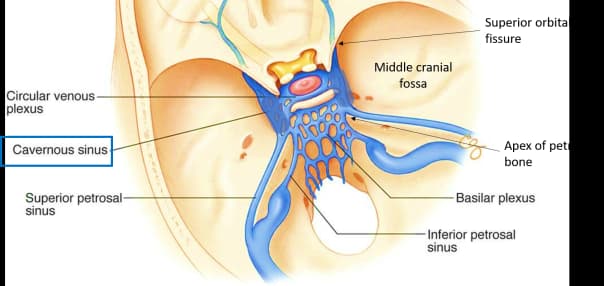
- Relations: The nasopharynx is immediately inferior to the skull base; tumours can erode superiorly into the cavernous sinus (containing CN III, IV, V1, V2, VI and the internal carotid artery) and the middle cranial fossa [2].
- Eustachian tube orifice opens here — this is why NPC causes unilateral serous otitis media.
- Boundaries: Extends vertically from the soft palate to the superior surface of the hyoid bone (floor of vallecula) [2]. Laterally bounded by the pharyngeal constrictor muscles and medial aspect of the mandible.
- Components: Tonsillar region, base of tongue, soft palate, posterolateral pharyngeal wall [2].
- Lymphatic drainage: Primarily to Level II (most common), then III, IV, V, parapharyngeal and retropharyngeal nodes [2]. Bilateral metastasis common from midline structures (tongue base, soft palate).
- This is the primary site for HPV-associated H&N cancer [2].
- Boundaries: From the level of the hyoid bone superiorly to the lower border of the cricoid cartilage inferiorly (where it becomes the oesophagus).
- Subsites: Pyriform sinus (most common subsite), postcricoid area, posterior pharyngeal wall.
- Tumours here tend to present late with advanced disease — the hypopharynx is capacious and tumours can grow large before causing symptoms.
- Associated with Paterson-Brown-Kelly syndrome (Plummer-Vinson syndrome) — triad of iron deficiency anaemia, dysphagia, and postcricoid web [3][2].
- Loss of laryngeal crepitus on examination is a sign of hypopharyngeal/postcricoid tumour (tumour fixes the larynx to the prevertebral fascia, abolishing the normal side-to-side glide of the larynx over the cervical spine) [3].
"Larynx" — from Greek larynx = throat/voice box.
Boundaries: Extends from the epiglottis superiorly to the cricoid cartilage inferiorly [2].
Three anatomical regions:
| Region | Boundaries | Clinical Features |
|---|---|---|
| Supraglottis | Epiglottis (tip to ventricular folds/false cords) | Presents as advanced disease due to paucity of symptoms; rich lymphatics → higher incidence of LN metastasis (30–50%) [2] |
| Glottis | True vocal cords + anterior and posterior commissures | Most common form of laryngeal cancer; presents early with hoarseness; limited lymphatics → low rate of nodal spread [2] |
| Subglottis | From 1 cm below free edge of vocal cord to inferior border of cricoid | Presents as advanced disease; propensity for local extension; higher recurrence rates; poorer survival [2] |
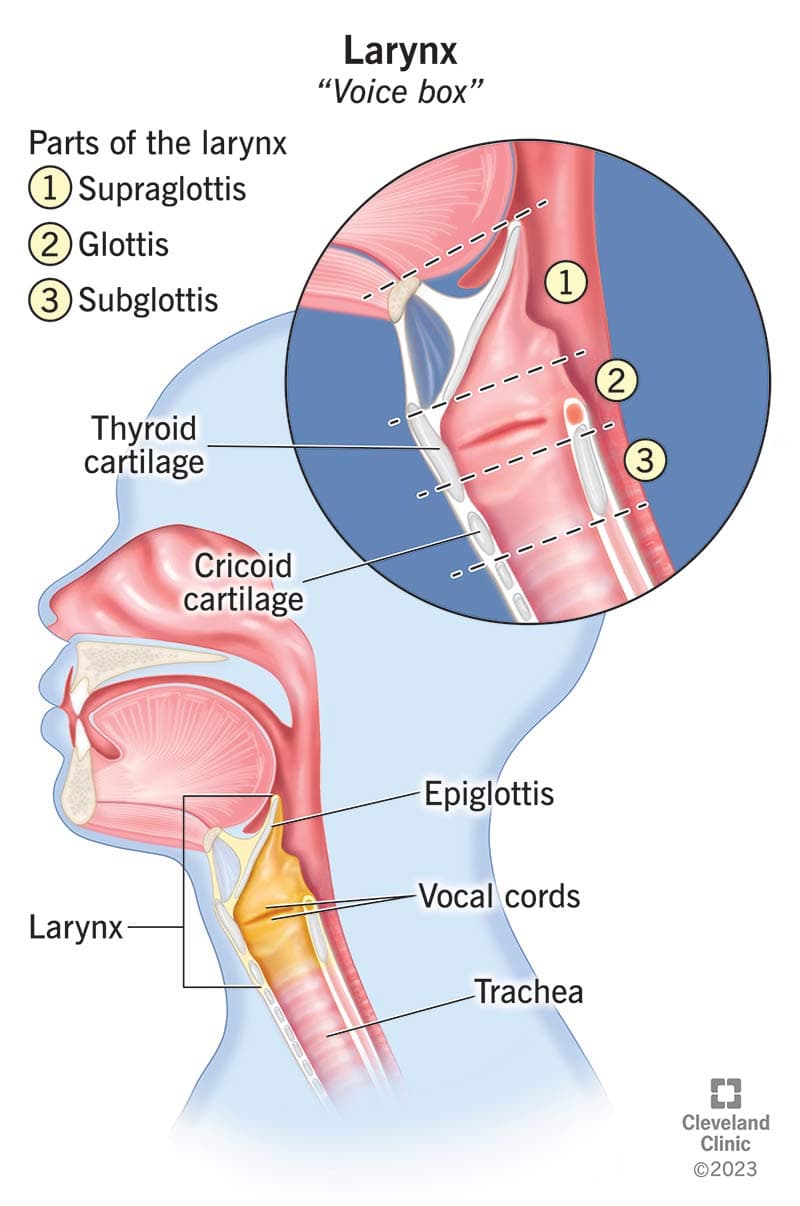
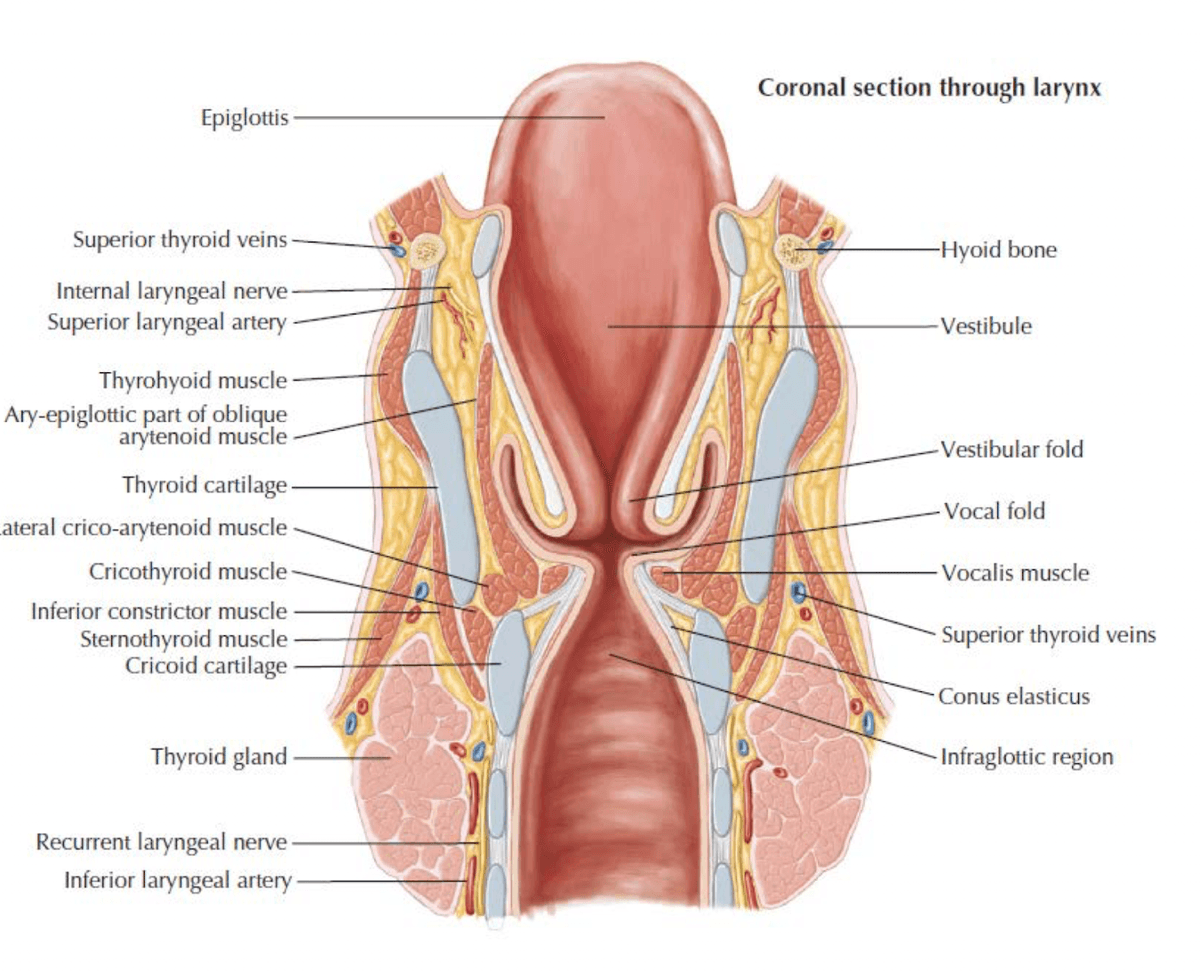
Functions of the larynx [2]:
- Phonation — production of a primary vocal tone at the level of the vocal folds. The sound resonates in the pharynx and nose (adding harmonics and timbre), then is articulated by fine motor control of the tongue, palate, and lips.
- Maintain airway patency and protection during swallowing — Normal swallow mechanism includes laryngeal elevation, posterior deflection of the epiglottis, inhibition of respiration, and closure of the vocal folds to prevent aspiration [2].
- Valsalva manoeuvre — Generation of increased pressure against a closed glottis. Enables coughing, throat clearing, straining, and defecation [2].
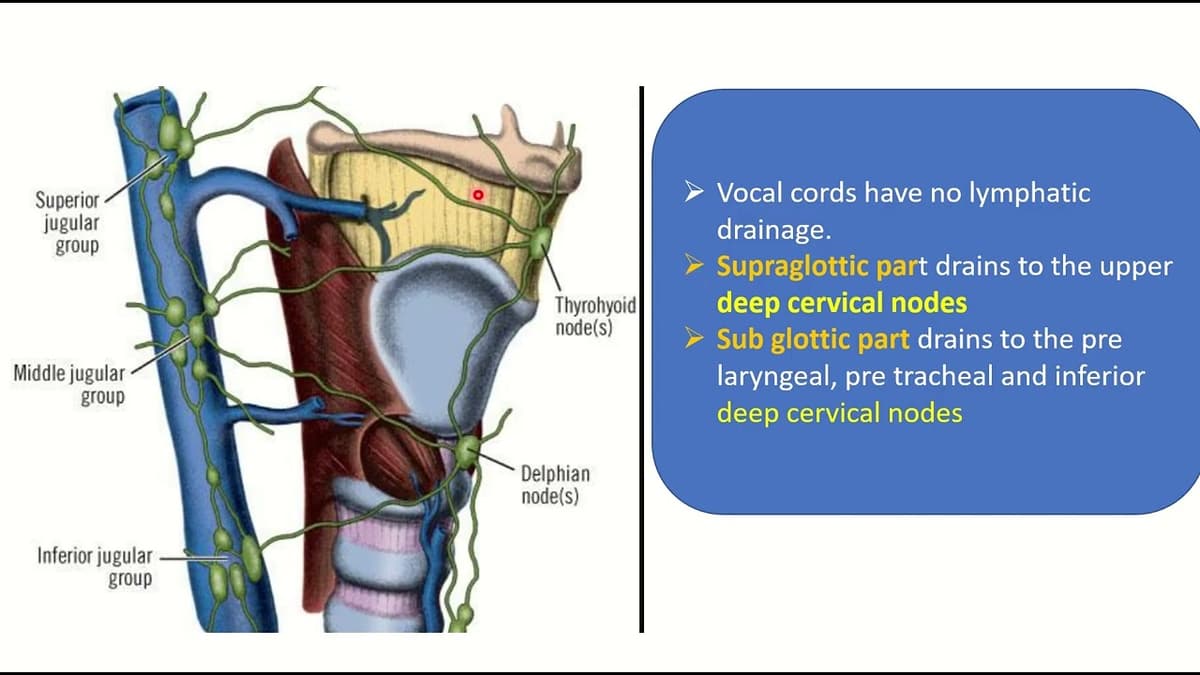
Lymphatic drainage [2]:
- Supraglottic: Pierces the thyrohyoid membrane with the superior laryngeal artery, vein, and nerve → drains to subdigastric and superior jugular nodes.
- Glottic and subglottic: Exits via cricothyroid ligament → prelaryngeal node (Delphian node), paratracheal nodes, and deep cervical nodes along the inferior thyroid artery.
- Sinuses: Maxillary, ethmoid, frontal, sphenoid.
- The maxillary sinus is the most common site for paranasal sinus malignancy.
- These cancers are rare but tend to present late because the sinus cavity allows silent tumour growth.
- Histology is more varied: SCC, adenocarcinoma (especially in woodworkers), adenoid cystic carcinoma, esthesioneuroblastoma (olfactory neuroblastoma), sinonasal undifferentiated carcinoma (SNUC).
- Major: Parotid (80% of salivary tumours), submandibular, sublingual.
- Minor: Scattered throughout the oral cavity and oropharynx (hard palate, tongue base, soft palate, etc.).
- Rule of thumb: The smaller the gland, the higher the proportion of malignancy.
- Parotid: ~20–25% malignant
- Submandibular: ~40–50% malignant
- Sublingual and minor: ~50–80% malignant
- Risk factors specific to salivary gland tumours include EBV (lymphoepithelial carcinoma), HPV (occasionally in mucoepidermoid carcinoma), HIV (increased incidence), radiation exposure, and smoking (Warthin's tumour is STRONGLY associated with smoking in contrast to other salivary gland tumours) [2].
4. Etiology and Risk Factors
The mnemonic to remember the 4 big factors for H&N cancer: HPV + EBV + Smoking + Alcohol [2]. An expanded mnemonic used is the "5 S's": Smoking, Spirits (alcohol), Sharp teeth (chronic trauma), Sex (male/oral sex), Spicy food [2].
Smoking is the PRIMARY risk factor for H&N cancer [3].
Pathophysiology: Tobacco smoke contains > 70 known carcinogens (e.g., polycyclic aromatic hydrocarbons, nitrosamines, aromatic amines). These cause:
- Direct DNA damage — adduct formation, point mutations (especially TP53 mutations in smoking-related HNSCC).
- Chronic mucosal irritation — leads to squamous metaplasia → dysplasia → carcinoma sequence.
- Impaired mucociliary clearance — prolonged contact time of carcinogens with mucosa.
Site predilection [2]:
- Tumours from smokers present more frequently in the floor of the mouth, hypopharynx, and larynx (areas of carcinogen pooling/contact).
- Tumours from non-smokers present more frequently in the oral cavity, especially the anterior tongue, buccal mucosa, and alveolar ridge.
Alcohol has a synergistic effect with smoking — the risk is multiplicative, not simply additive [2][3].
Pathophysiology:
- Solvent effect — alcohol acts as a solvent for tobacco carcinogens, enhancing mucosal penetration.
- Acetaldehyde — ethanol is metabolized to acetaldehyde (a known carcinogen) by alcohol dehydrogenase. Acetaldehyde causes DNA cross-links and point mutations.
- Nutritional deficiency — chronic alcoholism leads to folate, vitamin A, and zinc deficiency, impairing DNA repair and mucosal integrity.
- Particularly associated with hypopharyngeal carcinoma [3].
HPV infection, particularly types 16 and 18, is a major risk factor for oropharyngeal carcinoma [2][3].
Pathophysiology [2]:
- HPV infection induces two viral oncoproteins: E6 and E7
- E6 → binds and degrades p53 (tumour suppressor → normally triggers apoptosis in damaged cells). Without p53, damaged cells survive and accumulate mutations.
- E7 → binds and inactivates Rb (retinoblastoma protein → normally prevents cell cycle progression from G1 to S phase). Without Rb, cells undergo uncontrolled proliferation.
- This is a fundamentally different carcinogenic mechanism from smoking/alcohol (which cause direct DNA mutations in TP53). HPV tumours often have wild-type TP53 but it is functionally inactivated.
Clinical characteristics [2]:
- Presents in young male patients with a higher lifetime number of sexual partners and oral sex
- HPV-associated H&N cancer occurs primarily in the oropharynx, including tonsils and the base of tongue — the tonsillar crypt epithelium is particularly susceptible because it is a reticulated epithelium with gaps that allow HPV access to basal cells.
- Defines a distinct subset of patients compared with HPV-negative tobacco/alcohol-driven oropharynx cancer with:
- Frequent LN metastasis (often presents with a neck lump as the first symptom — can be cystic)
- Higher response rate to induction chemotherapy
- Better prognosis
- De-intensification of treatment can be considered while obtaining the same locoregional and overall survival [2]
HPV-positive vs HPV-negative Oropharyngeal Cancer
This distinction is so important that the AJCC 8th Edition (2017) TNM staging system has separate staging systems for HPV-positive (p16+) and HPV-negative oropharyngeal cancers. HPV-positive cancers are "downstaged" because they have a dramatically better prognosis (5-year survival ~80% vs ~50% for HPV-negative).
EBV is the primary etiological agent in the pathogenesis of NPC [2].
Pathophysiology [2]:
- EBV infects nasopharyngeal epithelial cells, where it can remain latent.
- Latent membrane protein 1 (LMP1) — acts as a constitutively active CD40 receptor, activating NF-κB, MAPK, and PI3K/Akt signalling pathways → promotes cell survival, proliferation, and immune evasion.
- EBNA1 — essential for EBV genome replication and maintenance in dividing cells.
- Detection of EBV DNA and EBV gene expression in precursor lesions and tumour cells [2].
- Serological responses: IgA antibodies against EBV viral capsid antigen (EBV VCA IgA) are elevated and used for screening/diagnosis [2].
- Plasma EBV DNA levels correlate with tumour burden and are used for monitoring treatment response and surveillance for recurrence (this is now a standard of care in HK).
Salted fish and preserved/fermented food — particularly relevant to NPC in Southern China [2].
Pathophysiology [2]:
- Contains high levels of nitrosamines, bacterial mutagens, direct genotoxins, and EBV-reactivating substances
- Cooking of salt-cured food releases volatile nitrosamines carried by steam and distributed over the nasopharyngeal mucosa
- These nitrosamines may also reactivate latent EBV in nasopharyngeal epithelial cells, creating a synergistic carcinogenic effect.
Associated with oral cavity carcinoma [3].
Pathophysiology:
- Areca nut contains arecoline — a parasympathomimetic alkaloid that is genotoxic and promotes oral submucous fibrosis (OSF), a premalignant condition.
- OSF → progressive fibrosis of the submucosal tissue → trismus and restricted mouth opening → malignant transformation in 7–13% of cases.
- Particularly prevalent in South and Southeast Asian populations (India, Taiwan, parts of mainland China).
- Family history of NPC is a risk factor [2].
- Associated with certain HLA haplotypes (e.g., HLA-A2, HLA-B46 in Southern Chinese populations) [2].
- Genetic polymorphisms such as CYP2A6 — a polymorphism of nitrosamine metabolizing gene. Certain variants metabolize nitrosamines less efficiently, leading to prolonged carcinogen exposure [2].
| Risk Factor | Details |
|---|---|
| Environmental UV light exposure | Primarily for lip cancer (lower lip). Projection of lower lip relating to sunlight exposure explains why the majority of SCC arise along the vermilion border of lower lip [2]. Also relevant for skin cancers of the H&N region. |
| Radiation exposure | Previous radiotherapy to H&N region (e.g., for childhood cancers, Hodgkin lymphoma, benign conditions). Latency of 10–20+ years [2]. |
| Immunosuppression | HIV, post-organ transplant, long-term immunosuppressive therapy [3]. |
| Poor oral hygiene with chronic infection | Chronic dental trauma, ill-fitting dentures (hard palate cancer, buccal mucosa cancer) [3][2]. |
| Chronic laryngitis / GERD / Laryngopharyngeal reflux | Risk factors for laryngeal carcinoma [2]. Chronic acid/pepsin exposure causes mucosal injury → metaplasia → dysplasia. |
| Previous malignancy | Field cancerization effect (see below) [3]. |
| Lichen planus | Oral lichen planus, especially erosive form, is a premalignant condition for oral SCC (buccal mucosa) [2]. |
| Plummer-Vinson syndrome | Triad of iron deficiency anaemia, dysphagia, and cervical oesophageal web. Well-established relationship with development of oral cancer and postcricoid/hypopharyngeal cancer [2]. |
| Subsite | Primary Risk Factors |
|---|---|
| Oral cavity | Smoking, alcohol, betel nut, chronic dental trauma, lichen planus, Plummer-Vinson syndrome |
| Oropharynx | HPV (especially types 16 and 18), smoking, alcohol, oral sex |
| Nasopharynx | EBV, salted fish/preserved food, genetics (HLA, CYP2A6), family history, smoking |
| Hypopharynx | Alcohol (synergistic with smoking), Plummer-Vinson syndrome |
| Larynx | Smoking, alcohol, chronic laryngitis, GERD/LPR, radiation, family history |
| Nasal cavity/paranasal sinuses | Woodworking (hardwood dust → adenocarcinoma), nickel refining, leather working, smoking |
| Salivary glands | Radiation, EBV (lymphoepithelial carcinoma), HIV, smoking (Warthin's tumour only) |
| Lip | UV light exposure, smoking, immunosuppression, fair complexion |
This is a critical concept — it explains why H&N cancer patients get second primary tumours (not metastases, but entirely new independent cancers).
Diffuse and chronic exposure of mucosa of the upper aerodigestive tract to carcinogenic substances leads to widespread changes in the mucosal epithelium [2].
Mechanism: Years of smoking and alcohol exposure cause genetic damage not just at the site of the primary tumour, but across the entire mucosal field. Multiple independent clones of pre-malignant cells develop in different locations. This leads to:
- Synchronous tumour = second primary tumour detected within 6 months [2]
- Metachronous tumour = second primary tumour detected > 6 months [2]
Clinical patterns [2]:
- Patients with oral cavity/oropharynx tumours → more likely to develop a second primary in the upper oesophagus (surveillance with chromoendoscopy, high-resolution white light endoscopy, or narrow band imaging (NBI) may be indicated)
- Patients with laryngeal tumours → more likely to develop a second primary in the lung
Implication [2]:
- Panendoscopy is ALWAYS recommended
- Panendoscopy includes direct laryngoscopy, bronchoscopy, and OGD
- Staging examination is recommended at the initial evaluation of ALL patients with primary cancers of the upper aerodigestive tract
Field Cancerization
A common exam mistake: confusing a second primary tumour with a metastasis. If a laryngeal cancer patient develops a new lung lesion, it may be a second primary lung cancer (from field cancerization) rather than a metastasis. The distinction matters because second primaries are potentially curable; metastases are usually not.
6. Classification
90% of H&N malignancies are squamous cell carcinoma (SCC) (not including nasopharynx and thyroid) [3].
| Histological Type | Typical Sites | Key Features |
|---|---|---|
| Squamous cell carcinoma | All sites (oral cavity, oropharynx, hypopharynx, larynx) | By far the most common. Graded as well, moderately, or poorly differentiated. |
| Non-keratinizing carcinoma | Nasopharynx | Most common endemic form of NPC (HK); undifferentiated subtype; strongly associated with EBV; more favourable prognosis [2] |
| Keratinizing SCC | Nasopharynx (sporadic form) | Most common sporadic form of NPC; resembles typical SCC; NOT strongly EBV-associated [2] |
| Basaloid SCC | Nasopharynx, oropharynx, hypopharynx | Aggressive clinical course; poor survival [2] |
| Verrucous carcinoma | Oral cavity, larynx | Well-differentiated, locally aggressive but rarely metastasizes. |
| Adenocarcinoma | Nasal cavity/paranasal sinuses, salivary glands | Minor salivary gland origin common. |
| Adenoid cystic carcinoma | Salivary glands (major and minor) | Perineural invasion is a hallmark. Slow but relentless. |
| Mucoepidermoid carcinoma | Parotid, minor salivary glands | Most common malignant salivary gland tumour. |
| Lymphoma | Tonsils and tongue base may be the presenting site for a lymphoma [2] | Think of this especially with a symmetrically enlarged tonsil. |
| Minor salivary gland tumours | May present as submucosal masses in the tongue base and soft palate [2] | Important differential for oropharyngeal masses. |
| WHO Type | Description |
|---|---|
| Non-keratinizing (undifferentiated) | Most common endemic form of NPC (HK). Strongly associated with EBV. More favourable prognosis. [2] |
| Keratinizing SCC | Most common sporadic form. Arises from squamous cells. Less EBV association. [2] |
| Basaloid SCC | Aggressive clinical course. Poor survival and prognosis. [2] |
The TNM staging differs by subsite. Key principles:
- T = size and extent of primary tumour (site-specific criteria)
- N = regional lymph node involvement
- M = distant metastasis
- Separate staging for HPV-positive (p16+) oropharyngeal cancer in AJCC 8th edition — reflects the dramatically better prognosis.
- NPC has its own distinct TNM staging — T stage based on anatomical extent (parapharyngeal extension, skull base invasion, intracranial extension, cranial nerve involvement).
These are clinically important because they represent opportunities for early detection and prevention.
| Lesion | Description | Malignant Potential |
|---|---|---|
| Leukoplakia | White patch or plaque that cannot be scraped off and cannot be characterized clinically or pathologically [2]. | Overall 3–5% malignant transformation. Leukoplakia on the floor of the mouth has a particularly high risk of malignant transformation [2]. |
| Erythroplakia | Bright red plaque of oral mucosa that cannot be characterized clinically or pathologically [2]. | Higher malignant potential than leukoplakia (~50% harbour dysplasia or carcinoma on biopsy). |
| Speckled leukoplakia | Variation of leukoplakia arising on an erythematous base [2]. | Highest rate of malignant transformation [2]. |
| Oral submucous fibrosis | Progressive fibrosis related to betel nut chewing. | 7–13% malignant transformation. |
| Oral lichen planus (erosive) | Chronic inflammatory condition of oral mucosa. | ~1–2% malignant transformation (controversial but well-recognized association). |
Premalignant Lesions: Red is Worse Than White
Erythroplakia (red) has a much higher malignant potential than leukoplakia (white). Speckled leukoplakia (mixed red and white) has the highest risk of all. If you see a red patch in the mouth — biopsy it.
7. Pathophysiology of Carcinogenesis
Normal mucosa → Hyperplasia → Mild dysplasia → Moderate dysplasia → Severe dysplasia/CIS → Invasive SCC
This is analogous to the adenoma-carcinoma sequence in colorectal cancer. Chronic carcinogen exposure (tobacco, alcohol) causes stepwise accumulation of genetic mutations:
- Early: p16/CDKN2A inactivation (cell cycle control)
- Intermediate: TP53 mutation (guardian of the genome)
- Late: Cyclin D1 amplification, EGFR overexpression
- In HPV-driven cancers: p53 and Rb are functionally inactivated by E6/E7 rather than mutated.
| Pathway | Relevance |
|---|---|
| TP53 | Mutated in ~60–80% of smoking-related HNSCC. Loss of apoptosis and cell cycle arrest. |
| Rb | Inactivated by HPV E7 or mutated in a subset of HNSCC. Loss of G1/S checkpoint. |
| EGFR | Overexpressed in ~90% of HNSCC. Drives proliferation via Ras/MAPK and PI3K/Akt. Target of cetuximab. |
| PI3K/Akt/mTOR | Frequently activated in HNSCC. Target of ongoing clinical trials. |
| PD-L1 | Immune checkpoint often upregulated in HNSCC. Target of pembrolizumab/nivolumab (immunotherapy). |
| NF-κB | Activated by EBV LMP1 in NPC. Promotes survival and immune evasion. |
8. Clinical Features
The clinical presentation of H&N cancer depends on the subsite and extent of the primary tumour. The key is to think anatomically — where is the tumour? — and then work out what structures it affects.
History taking should systematically cover [3]:
- Age, Sex
- Duration: Acute vs Chronic
- Symptoms (see below, organ by organ)
- Risk factors: Smoking, alcohol, family history
- Functional disturbances: Breathing, chewing, swallowing, phonation, articulation
- Co-morbidities
8.2 Symptoms by Organ System
- Unilateral hearing loss [3] — conductive hearing loss from serous otitis media (middle ear effusion). The tumour obstructs the Eustachian tube orifice in the nasopharynx → negative middle ear pressure → fluid accumulation. This is the classic NPC presentation.
- Rule: Any adult in Southern China with new-onset unilateral serous otitis media must have the nasopharynx examined to rule out NPC.
- Otalgia (ear pain) [3] — can be direct (NPC invading the ear) but more commonly is referred otalgia. Why? The pharynx is innervated by CN IX (glossopharyngeal) and CN X (vagus), which also supply sensory branches to the ear (Jacobson's nerve from CN IX; Arnold's nerve from CN X). So an oropharyngeal, hypopharyngeal, or tongue base cancer can present with ear pain — the ear itself is normal.
Referred Otalgia
If a patient has persistent unilateral ear pain and the ear looks normal on otoscopy, you MUST examine the oropharynx, hypopharynx, and larynx. Referred otalgia from a H&N cancer is a commonly missed diagnosis.
- Blood-stained nasal discharge [3] — NPC or sinonasal tumour eroding into mucosal blood vessels.
- Unilateral nasal obstruction [3] — tumour mass blocking the nasal airway. Unilateral is the key word — bilateral obstruction is more likely inflammatory (e.g., allergic rhinitis). Unilateral, especially with bloody discharge, is cancer until proven otherwise.
- Non-healing ulcers [3] — the hallmark of oral cavity SCC. Any oral ulcer that does not heal within 3 weeks should be biopsied.
- Mass [3] — may be exophytic (outward-growing) or endophytic (infiltrative).
- Blood-stained saliva [3] — tumour surface bleeds with minimal trauma.
- Loosening of denture [3] — tumour growth beneath the denture or destruction of the alveolar ridge.
- Pain/soreness — may be surprisingly mild initially; becomes severe with deep invasion.
- Paraesthesia — numbness in the lip (mental nerve involvement from mandibular tumour), tongue (lingual nerve involvement from tongue or floor of mouth cancer), or face (infraorbital nerve involvement from maxillary sinus cancer).
- Ipsilateral paraesthesia of tongue → invasion of lingual nerve [2].
- Tongue deviation, fasciculation, and atrophy → invasion of hypoglossal nerve (CN XII) by locally extensive tumours [2]. The tongue deviates towards the side of the lesion (because the denervated genioglossus can no longer push the tongue to the contralateral side).
- Trismus — inability to fully open the mouth. Caused by invasion into the medial pterygoid muscle or involvement of the ascending ramus of mandible [2]. This is a sign of advanced disease.
- Hoarseness [3] — the cardinal symptom of glottic laryngeal cancer (tumour on the vocal cord impairs vibration → dysphonia). Can also occur from recurrent laryngeal nerve (RLN) invasion (from thyroid, oesophageal, or lung apex cancer causing vocal cord paralysis). Any hoarseness lasting > 3 weeks requires laryngoscopy.
- Blood-stained sputum [3] — haemoptysis from laryngeal or hypopharyngeal tumour.
- Shortness of breath / stridor [3] — late symptom indicating significant airway narrowing by tumour mass. Stridor (high-pitched inspiratory noise) = > 50% airway occlusion. This is an emergency.
- Muffled ("hot potato") voice — seen in oropharyngeal tumours (especially tonsillar) due to mass effect on the pharyngeal resonating space [3].
- Globus (sensation of a lump in the throat) [3] — early symptom of hypopharyngeal cancer. Non-specific but should prompt investigation in high-risk patients.
- Dysphagia [3] — difficulty swallowing. Progressive dysphagia from solids to liquids suggests an obstructive lesion. Can also result from reduced tongue mobility (tongue cancer), trismus, or pharyngeal wall invasion.
- Odynophagia (painful swallowing) [3] — indicates mucosal ulceration or deep invasion.
- Sore throat [3] — persistent, unilateral sore throat in an adult is a red flag.
- Cervical lymphadenopathy [3] — may be the first and only presenting symptom, particularly in:
- NPC (bilateral, posterior triangle)
- HPV-positive oropharyngeal cancer (often cystic Level II nodes)
- Occult primary (unknown primary with neck node metastasis)
- 50% of oropharyngeal cancers have cervical LN metastasis at presentation [2][3].
- 30% of hypopharyngeal cancers have LN metastases [3].
- Weight loss — common in advanced disease due to cancer cachexia and mechanical difficulty eating/swallowing.
- Fever, night sweats — think lymphoma if these are prominent [2].
| Subsite | Early Symptoms | Late/Advanced Symptoms |
|---|---|---|
| Lip | Non-healing ulcer on vermilion border | Paraesthesia (mental nerve) |
| Oral tongue | Non-healing ulcer, pain | Tongue deviation (CN XII), ipsilateral tongue paraesthesia (lingual nerve), speech difficulty |
| Floor of mouth | Ulcer, pain | Trismus, submandibular swelling |
| Buccal mucosa | Ulcer, white/red patch | Trismus (pterygoid invasion) |
| Hard palate | Mass, ill-fitting denture | Oro-antral fistula (palate perforation) |
| Oropharynx | Sore throat, referred otalgia, dysphagia/odynophagia, muffled speech [3] | Mass/ulcer, trismus, asymmetrical tonsil, cervical LN (50%) [3] |
| NPC | Unilateral serous otitis media, epistaxis, nasal obstruction | Cranial nerve palsies (III–VI), proptosis, neck lump (bilateral) |
| Hypopharynx | Sore throat, globus → dysphagia, otalgia, hoarseness [3] | Loss of laryngeal crepitus, 30% LN metastases [3] |
| Supraglottic larynx | Vague sore throat, referred otalgia, globus | Hoarseness (extension to glottis), dysphagia, neck lump, airway obstruction |
| Glottic larynx | Hoarseness (early!) | Airway obstruction, stridor, dysphagia |
| Subglottic larynx | Often silent | Stridor, dyspnoea, haemoptysis |
8.4 Signs on Examination
- Nutritional status — cachexia in advanced disease.
- Voice quality — hoarse (glottic), muffled/hot potato (oropharyngeal), hypernasal (palatal defect), hyponasal (nasal obstruction).
- Trismus — measure interincisal distance (normal > 40mm; < 35mm is significant trismus).
- Skin changes — radiation dermatitis (if previously treated), solar keratosis (lip cancer).
- Non-healing ulcer — indurated (hard) edges, raised/rolled margins, contact bleeding.
- Exophytic mass — fungating, friable.
- Submucosal mass — suggests minor salivary gland tumour or deep-seated tumour.
- Leukoplakia/erythroplakia/speckled leukoplakia — premalignant.
- Floor of mouth — bimanual palpation (finger in mouth + finger under chin) to assess induration.
- Lymphadenopathy — systematically examine all neck levels (I–VI).
- Hard, fixed, non-tender nodes suggest metastatic carcinoma.
- Rubbery, mobile nodes suggest lymphoma.
- Cystic nodes in a young patient → think HPV-positive oropharyngeal cancer (cystic metastasis).
- Loss of laryngeal crepitus [3] — normally, the larynx can be rocked side to side over the cervical spine with a palpable crepitus. Loss of this sign indicates postcricoid or hypopharyngeal tumour fixing the larynx to the prevertebral fascia.
NPC can invade the skull base and affect multiple cranial nerves. Systematic CN examination is essential.
| CN Affected | Clinical Sign | Mechanism |
|---|---|---|
| CN III, IV, VI | Diplopia, ptosis, squint | Invasion of cavernous sinus [2] |
| CN V (V1, V2, V3) | Facial numbness, paraesthesia | Skull base erosion, cavernous sinus invasion |
| CN V3 (motor) | Weakness of mastication muscles | Foramen ovale invasion |
| CN IX | Dysphagia, loss of gag reflex | Parapharyngeal space invasion |
| CN X | Hoarseness (vocal cord paralysis) | Parapharyngeal space, vagal nerve involvement |
| CN XII | Tongue deviation, atrophy | Hypoglossal canal invasion |
| Sympathetic chain | Horner's syndrome (miosis, ptosis, anhidrosis) | Parapharyngeal space invasion |
- Asymmetrical tonsil [3] — suggests tonsillar carcinoma or lymphoma. An asymmetrically enlarged tonsil in an adult should be biopsied.
- Mass in Fossa of Rosenmüller — NPC.
- Vocal cord mobility — immobile cord = invasion of RLN or cricoarytenoid joint fixation.
- Pyriform sinus pooling — indirect sign of hypopharyngeal obstruction.
9. Patterns of Metastasis
Understanding lymphatic drainage patterns is essential for predicting nodal spread and planning surgery.
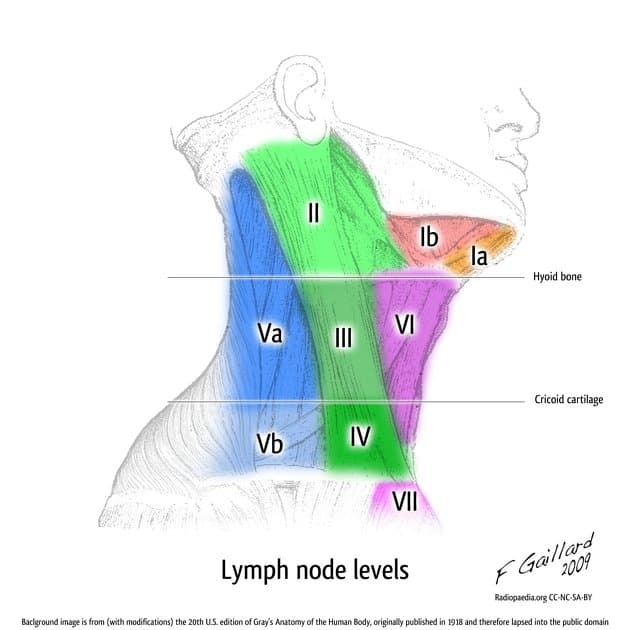
| Level | Location | Primary Drainage From |
|---|---|---|
| I | Submental (IA), Submandibular (IB) | Lip, anterior oral cavity, floor of mouth |
| II | Upper jugular (jugulodigastric) | Oral cavity, oropharynx, nasopharynx, supraglottic larynx |
| III | Mid-jugular | Oropharynx, hypopharynx, larynx |
| IV | Lower jugular | Hypopharynx, larynx, thyroid, oesophagus |
| V | Posterior triangle | Nasopharynx, oropharynx, skin of posterior scalp |
| VI | Central compartment (pretracheal, paratracheal) | Larynx (subglottic), thyroid |
| VII | Superior mediastinal | Thyroid, oesophagus |
| Retropharyngeal | Behind pharynx | Nasopharynx, oropharynx, hypopharynx |
- SCC of oral cavity and lips tends to metastasize to Level I, II, and III [2]
- SCC of the tongue tends to skip metastasize to Level III and IV [2] — this is why even early tongue cancer may need a neck dissection.
- Oropharyngeal cancer metastasizes to Level II (most common), III, IV, V, parapharyngeal and retropharyngeal and contralateral nodal groups [2]
- Bilateral LN metastasis common from tumours arising in the tongue base and soft palate [2] — because midline structures have bilateral lymphatic drainage.
- High incidence of lymphatic spread from supraglottic (30–50%) and subglottic cancer (40%) but limited glottic cancer typically does not spread to regional lymphatics [2] — the true vocal cord has sparse lymphatic drainage (limited to a superficial network), which is why early glottic cancer is rarely nodal.
- NPC: Lymph node metastases usually present at diagnosis and are commonly bilateral [2]. Drainage to retropharyngeal nodes and Level II–V.
10. Specific Subtypes: Key Clinical Features
- Majority diagnosed in the lower lip (88–98%) followed by upper lip (2–7%) and oral commissure (1%) [2].
- Lower lip predominance explained by the projection of the lower lip relating to sunlight exposure — SCC arises along the vermilion border [2].
- Clinical presentation: Ulcerated lesion on vermilion or cutaneous surface [2]. Paraesthesia in area adjacent to lesion indicates mental nerve involvement [2].
- LN metastasis: < 10%, primarily to submental and submandibular lymph nodes (Level I) [2].
- Risk factors: prolonged sunlight exposure, smoking, immunosuppression, fair complexion [2].
- Tumours occur most commonly on the lateral and ventral surfaces [2] — these are the areas of greatest contact with pooled saliva (which concentrates tobacco carcinogens).
- Early features: Non-healing ulcer, exophytic growth.
- Advanced features: Ipsilateral paraesthesia of tongue (lingual nerve invasion) [2]. Tongue deviation, fasciculation, and atrophy (hypoglossal nerve invasion) [2].
- Second most common oral cavity cancer subsite (after tongue).
- Leukoplakia on the floor of the mouth has a particularly high risk of malignant transformation [2].
- Early invasion of mandible (the periosteum of the mandible is thin here).
- Frequently originates from the pharyngeal recess (Fossa of Rosenmüller) [2]
- Remains asymptomatic for a long period due to its presence in a clinically occult site [2]
- Patient will present with locally or regionally advanced disease due to prolonged asymptomatic period or missed diagnosis [2]
- Tendency for early metastasis [2]
- Classic presentation: Trotter's triad — unilateral conductive hearing loss + ipsilateral facial pain (CN V) + ipsilateral palatal paralysis.
- Male predominance (M:F = 2–3:1) [2]
- Glottic larynx: MOST common form; presents with hoarseness → diagnosed early [2]
- Supraglottic: presents as advanced disease; rich lymphatics → high nodal rate [2]
- Subglottic: presents late; propensity for local extension; poorer survival [2]
- Male predominance reflecting effect of smoking and alcohol use [2]
H&N cancers uniquely affect critical human functions — this is what makes these cancers particularly devastating. The lecture specifically emphasises:
Functional disturbances to assess [3][5]:
- Breathing — airway obstruction, tracheostomy dependency
- Chewing — trismus, loss of teeth/mandible, reduced tongue mobility
- Swallowing — dysphagia, aspiration risk, PEG tube dependency
- Phonation — hoarseness, voice loss (after laryngectomy)
- Articulation — impaired speech intelligibility after tongue/palate resection
Head and neck cancer problems relate to both function and shape [5]:
- Function: The structures involved (tongue, palate, larynx, mandible) are responsible for airway, swallowing, speech, and facial expression.
- Shape: The face and neck are cosmetically visible — disfigurement from disease or treatment causes profound psychosocial impact.
Function and Shape — The Dual Challenge
H&N cancer is unique because both the disease and its treatment can devastate essential human functions (breathing, eating, speaking) AND cosmetic appearance. Every treatment decision must balance oncological outcome against functional and cosmetic consequences. [5]
Surgery may cure your cancer — but the principles must be adhered to [6].
Key concepts from surgical oncology:
- Adequate surgical margin — the goal is to achieve negative margins (R0 resection). In H&N cancer, a margin of ≥ 5mm (ideally ≥ 10mm for oral cavity SCC) is considered adequate. Positive margins (R1) or close margins (< 5mm) are indications for re-excision or postoperative radiotherapy [6].
- Frozen section — intraoperative pathological assessment of margins. Allows real-time decision-making.
- Reconstruction after resection — H&N surgery often requires complex reconstruction:
- Reconstruction using soft pliable fasciocutaneous free flaps can provide intraoral bulk and preservation of tongue mobility [2]
- Prosthetic augmentation can allow contact between remaining tongue tissue and palate, improving ability to speak and swallow [2]
- Through-and-through defects of palate require dental prosthesis for rehabilitation of swallowing and speech [2]
- Neck dissection — systematic removal of lymph nodes from specified levels:
- Selective neck dissection — removes only specified levels at risk
- Modified radical neck dissection — removes Levels I–V, preserving one or more of: spinal accessory nerve (CN XI), internal jugular vein (IJV), sternocleidomastoid (SCM)
- Radical neck dissection — removes Levels I–V + CN XI + IJV + SCM (rarely performed now)
High Yield Summary
Definition: H&N cancers are predominantly SCC arising from the mucosal surfaces of the UADT — oral cavity, pharynx (naso-, oro-, hypo-), larynx, nasal cavity/paranasal sinuses, and salivary glands.
Epidemiology: Male predominance, age > 60 (except HPV-related oropharyngeal cancer in younger males). NPC is endemic in Southern China/Hong Kong.
4 Major Risk Factors: HPV (oropharynx) + EBV (NPC) + Smoking + Alcohol. Mnemonic: 5 S's — Smoking, Spirits, Sharp teeth, Sex (male/oral), Spicy food.
Field Cancerization: Diffuse carcinogen exposure → synchronous/metachronous tumours. Always do panendoscopy (direct laryngoscopy + bronchoscopy + OGD).
HPV Mechanism: E6 degrades p53, E7 inactivates Rb → better prognosis, de-intensification possible.
EBV Mechanism: LMP1 activates NF-κB → NPC. Plasma EBV DNA used for screening/monitoring.
Premalignant Lesions: Erythroplakia > Speckled leukoplakia > Leukoplakia in malignant potential.
Key Presentations by Site:
- Lip: Non-healing ulcer on vermilion border (UV exposure)
- Oral tongue: Lateral/ventral ulcer; lingual nerve (paraesthesia), CN XII (deviation)
- NPC: Unilateral serous otitis media, epistaxis, cranial nerve palsies, bilateral neck nodes
- Oropharynx: Sore throat, referred otalgia, dysphagia, muffled voice, 50% cervical LN
- Hypopharynx: Globus → dysphagia, otalgia, hoarseness, loss of laryngeal crepitus
- Glottic larynx: Early hoarseness (most common laryngeal cancer, best prognosis)
- Supraglottic/Subglottic: Late presentation, worse prognosis
Lymphatic Drainage: Glottic = sparse (low nodal risk); Supraglottic = rich (30–50% nodal); Tongue = skip metastasis to Level III–IV.
Always protect the airway.
Active Recall - Head and Neck Cancer (Definition to Clinical Features)
[1] Senior notes: felixlai.md (H&N cancer overview, Section I) [2] Senior notes: felixlai.md (H&N cancer, NPC, CA Oropharynx, Laryngeal carcinoma, Lip/Tongue/Buccal/Palate cancers, Salivary gland tumours sections) [3] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (pp. 8, 37, 39, 40, 41) [4] WHO Global Cancer Observatory (GLOBOCAN 2022) [5] Lecture slides: GC 187. Head and neck cancer problems Function and shape.pdf [6] Lecture slides: GC 202. Surgery may cure your cancer Surgical oncology.pdf [7] Image credit: Cleaveland Clinic (pharynx image) [8] Image credit: Standard of Care. https://standardofcare.com/pharyngeal-recess/ [9] Image credit: Anatomy QA [10] Image credit: Cleavelenad clnnic (larynx anatomy image) [11] Image credit: Radiopaedia.org (lymph node levels image)
Differential Diagnosis of Head and Neck Cancer
The differential diagnosis of a head and neck mass or mucosal lesion is broad. The critical clinical skill is distinguishing malignant from benign and inflammatory conditions — because the management is radically different. Let's think about this systematically, starting from first principles: What can cause a lump or lesion in the head and neck?
The answer falls into three fundamental categories: Congenital/Developmental, Inflammatory/Infective, and Neoplastic. The lecture slides give us a crucial clinical pearl for distinguishing these:
Clinical presentations of oral cavity and oropharyngeal conditions [3]:
- Infective: acute and febrile
- Neoplastic (congenital/developmental/malignant): chronic and afebrile
This is your first-pass filter on the ward round. A patient with a 3-day history of painful throat swelling and fever almost certainly has an infection. A patient with a 3-month, painless, progressive mass is neoplastic until proven otherwise.
1. Structured Approach: DDx by Presentation
The way a H&N cancer presents determines your differential diagnosis. Let's organise this by the presenting complaint.
This is the most common presentation of oral cavity and oropharyngeal cancer. You see a lesion in the mouth — what else could it be?
| Category | Differentials | Key Distinguishing Features |
|---|---|---|
| Malignant | SCC (by far most common — 90% of H&N malignancies [3]) | Indurated, non-healing ulcer > 3 weeks, contact bleeding, associated leukoplakia/erythroplakia |
| Adenocarcinoma (minor salivary gland origin) | Smooth submucosal mass, often hard palate or tongue base [3] | |
| Lymphoma | Tonsils and tongue base may be the presenting site for a lymphoma [2]. Rubbery, non-ulcerated tonsillar enlargement; may have B-symptoms | |
| Minor salivary gland tumours | May present as submucosal masses in the tongue base and soft palate [2]. Smooth, well-circumscribed, submucosal — unlike the irregular surface of SCC | |
| Verrucous carcinoma | Exophytic, warty, well-differentiated; locally aggressive but rarely metastasizes | |
| Mucosal melanoma | Pigmented lesion, very rare in oral cavity | |
| Kaposi's sarcoma | Violaceous flat/raised lesions; associated with HIV/HHV-8 | |
| Premalignant | Leukoplakia | White patch that cannot be scraped off [2]. Overall 3–5% malignant transformation |
| Erythroplakia | Bright red plaque [2]. ~50% harbour dysplasia/carcinoma on biopsy | |
| Speckled leukoplakia | Highest rate of malignant transformation [2] | |
| Oral submucous fibrosis | Betel nut chewing; trismus; 7–13% transformation | |
| Benign neoplasm | Fibroma | Most common benign oral tumour. Smooth, pedunculated, painless |
| Papilloma | Pedunculated, finger-like projections; HPV-related | |
| Granular cell tumour | Firm submucosal nodule, often tongue | |
| Pleomorphic adenoma of minor salivary gland | Smooth, slow-growing, hard palate | |
| Infective | Aphthous ulcer | Recurrent, painful, < 2cm, self-limiting (< 2 weeks). Not indurated. |
| Herpetic stomatitis | Vesicles → shallow ulcers, clustered, painful, self-limiting | |
| Oral candidiasis (thrush) | White plaques that CAN be scraped off (unlike leukoplakia) | |
| Syphilitic chancre (primary) | Painless ulcer with raised rolled edges — can mimic SCC! Serological testing essential | |
| Tuberculous ulcer | Chronic, painful ulcer with undermined edges; granulomatous histology | |
| Deep fungal infection (histoplasmosis) | Rare; immunocompromised | |
| Inflammatory | Traumatic ulcer | History of trauma (biting, sharp tooth, denture); resolves when cause removed |
| Lichen planus (erosive) | Wickham's striae, bilateral, chronic; premalignant | |
| Behçet's disease | Recurrent oral + genital ulcers, uveitis | |
| Pemphigus vulgaris | Flaccid blisters → widespread ulceration, positive Nikolsky's sign |
The 3-Week Rule
Any oral ulcer that does not heal within 3 weeks must be biopsied to rule out malignancy. This is a fundamental clinical rule. The lecture emphasises: Persistent 2–4 weeks after conservative/empirical treatment should trigger EARLY REFERRAL to ENT Surgeons when suspecting malignancy [3].
A neck mass is one of the most common presentations of H&N cancer — either from the primary tumour itself (e.g., salivary gland, thyroid) or from lymph node metastasis (the primary is elsewhere in the UADT). The differential is organised by location and patient demographics.
By Location [2]:
| Location | Differentials |
|---|---|
| Midline (Central) | Thyroid nodule (isthmus), Thyroglossal duct cyst, Dermoid cyst, Ranula, Level I lymph node [2] |
| Anterior triangle | Thyroid nodule, Branchial cleft cyst, Carotid body tumour (Chemodectoma), Carotid artery aneurysm, Laryngocoele, Lymphadenopathy (Levels I–IV) [2] |
| Posterior triangle | Lymphadenopathy (Level V), Cystic hygroma, Lipoma, Cervical rib |
By Age Group:
| Age | Most Likely Cause |
|---|---|
| Children (< 15) | Congenital (thyroglossal cyst, branchial cleft cyst, dermoid), Reactive lymphadenopathy, Lymphoma |
| Young adults (15–40) | Reactive lymphadenopathy, Branchial cleft cyst, Lymphoma, Salivary gland tumour, Thyroid nodule |
| Adults (> 40) | Metastatic carcinoma (most common cause of a neck mass in adults > 40 with risk factors), Lymphoma, Salivary gland tumour, Thyroid carcinoma |
By Pathological Category [2]:
| Category | Differentials | Key Features |
|---|---|---|
| Metastatic H&N carcinoma | Metastatic SCC from UADT (most common) [2] | Hard, fixed, non-tender. Masses are usually asymptomatic but symptoms related to the primary site can be elicited [2] — e.g., hoarseness, dysphagia, otalgia → cervical LN metastasis from an underlying UADT malignancy [2] |
| Thyroid masses | Thyroid nodule/cyst/carcinoma [2] | Moves with swallowing. Confirmed with ultrasound and FNA [2] |
| Salivary gland tumour | 80% arise in parotid gland; parotid tumours usually benign (80%); submandibular gland tumours usually malignant (50%) [2] | Tail of parotid or submandibular triangle location |
| Lymphoma | Hodgkin and Non-Hodgkin lymphoma | Neck involvement common in children with Hodgkin lymphoma (80%). Presents with fever, night sweats, chills and diffuse lymphadenopathy [2]. Rubbery, non-tender |
| Paraganglioma | Carotid body tumour (Chemodectoma) [2] | Pulsatile, bruit on auscultation, mobile side-to-side but not up-and-down (Fontaine's sign) [2]. Highly vascular. |
| Jugulotympanic paraganglia (Glomus jugulare) [2] | Pulsatile tinnitus, conductive hearing loss | |
| Schwannoma | Vagus nerve or sympathetic chain schwannoma [2] | Vagal schwannoma → hoarseness/aspiration; Sympathetic chain schwannoma → Horner's syndrome [2] |
| Congenital | Branchial cleft cyst (2nd most common; anterior to SCM) [2] | Presents in late childhood/early adulthood when infected. |
| Thyroglossal duct cyst | Midline, moves with swallowing AND tongue protrusion (distinguishes from thyroid nodule which moves with swallowing only) | |
| Dermoid cyst | Midline, submental, doughy feel | |
| Cystic hygroma (lymphatic malformation) | Transilluminant, posterior triangle, infants | |
| Benign | Lipoma [2] | Soft, ill-defined, slowly enlarging, any location [2] |
| Benign skin cysts (epidermoid, pilomatrixoma) [2] | Superficial, mobile | |
| Vascular | Carotid artery aneurysm | Pulsatile, expansile |
Cystic Neck Mass in a Young Adult — Don't Be Fooled
A cystic Level II neck mass in a young adult (especially a non-smoker) is classically thought to be a branchial cleft cyst. However, HPV-positive oropharyngeal SCC metastatic to Level II nodes can present as a cystic neck mass — the metastasis undergoes cystic degeneration. Always examine the oropharynx (tonsils and tongue base) and consider biopsy/FNA for p16 and HPV testing before excising what you think is a "branchial cleft cyst" in anyone over 30.
Different subsites have specific differentials beyond SCC:
Oral cavity [3]:
- Histology considerations — Epithelium (ulcerative): SCC / Adenocarcinoma [3]
- Underlying structure (smooth): Lymphoma / Minor salivary gland tumours [3]
Oropharynx [3]:
Nasopharynx:
- Non-keratinizing carcinoma (undifferentiated, EBV-associated) — dominant histology in HK [2]
- Lymphoma — nasopharyngeal lymphoma (NK/T cell type) is particularly relevant in Asian populations
- Adenoid cystic carcinoma (rare)
- Juvenile nasopharyngeal angiofibroma — benign but locally aggressive, exclusively in adolescent males. Highly vascular — DO NOT biopsy before imaging!
- Thornwaldt's cyst — benign notochordal remnant in nasopharyngeal midline
- Rathke's pouch cyst — developmental
Larynx:
- SCC (most common malignancy) [2]
- Vocal cord polyp/nodule — benign, related to voice abuse; sessile or pedunculated, usually unilateral
- Papilloma — can be recurrent respiratory papillomatosis (HPV 6, 11); mainly children
- Contact granuloma — posterior glottis, related to reflux or intubation trauma
- Reinke's oedema — polypoid degeneration of vocal cords (smoking, voice abuse)
- Laryngeal cyst (retention cyst, saccular cyst)
- Chondroma / Chondrosarcoma — rare, arising from laryngeal cartilage
Salivary glands [2]:
- Differential diagnosis of parotid/salivary mass:
- Salivary cysts
- Salivary gland stones
- Sjögren's syndrome
- Metastasis from other tumours (scalp/facial skin SCC or melanoma)
- Lymphoepithelial cysts (HIV-associated)
- Chronic sclerosing sialadenitis (Küttner's tumour)
- Regional lymphadenopathy (intraparotid lymph nodes)
- Sialadenosis — non-inflammatory, non-neoplastic hypertrophy; associated with anorexia/bulimia nervosa, alcoholic cirrhosis, diabetes mellitus [2]
3. Key Differentials Requiring Special Attention
Both can present as an asymmetric tonsillar mass. How do you tell them apart?
| Feature | SCC | Lymphoma |
|---|---|---|
| Surface | Ulcerated, irregular, friable | Smooth, intact mucosa (submucosal mass) |
| Consistency | Hard, indurated | Rubbery, firm |
| Unilateral vs bilateral | Usually unilateral | Can be bilateral |
| Systemic symptoms | Rare (weight loss late) | Fever, night sweats, chills [2] |
| LN involvement | Regional (Level II) | Diffuse, may be widespread |
| Definitive test | Incisional biopsy | Excisional/core biopsy (architecture needed for subtyping) |
Biopsy Approach Matters
FNA does NOT provide material for tissue architecture or immunohistochemical analysis [2]. For suspected lymphoma, you need a core needle biopsy or excisional biopsy for tissue architecture — FNA cytology alone cannot subtype lymphoma. This is a common exam question.
This is a classic clinical scenario: a patient presents with a neck lump, FNA shows metastatic SCC, but you cannot find the primary tumour on clinical examination.
Approach:
- Panendoscopy with biopsy — direct laryngoscopy + bronchoscopy + OGD + directed biopsies of likely primary sites (nasopharynx, tongue base, tonsils, pyriform sinus) [3]
- Tonsillectomy or EUA + Bx [3] — the tonsils are the most common site of occult primary (especially HPV-positive SCC hiding within tonsillar crypts)
- CT/MRI for anatomical delineation [3]
- PET scan — PET scan is superior to both CT and MRI for detecting regional nodal metastasis as well as distant metastasis and second primary tumours [2]
- FNA for p16/HPV testing and EBV (plasma EBV DNA) [2]
Facial weakness in the context of a parotid mass is high suspicion of malignant involvement of parotid gland [2]. Benign parotid tumours (even large ones) almost never cause facial nerve palsy because they displace rather than invade the nerve. If CN VII is affected, the lesion is likely malignant.
Other features suggesting malignancy:
- Rapid growth
- Pain and paraesthesia [2]
- Fixation to skin or deep structures
- Mucosal ulceration [2]
- Cervical lymphadenopathy
Parotid tumour must be distinguished from Bell's palsy [2] — both cause facial weakness, but Bell's palsy is acute onset, involves the entire hemifacial musculature equally (forehead included — lower motor neurone pattern), and has no palpable mass.
Differential diagnosis of bilateral parotid gland enlargement [2]:
- Parotitis (viral — mumps; bacterial — ascending infection)
- Bruxism (excess teeth grinding or jaw clenching — masseter hypertrophy can mimic parotid enlargement)
- Masseter hypertrophy
- Sialadenosis — non-inflammatory, non-neoplastic hypertrophy, usually bilateral and painless. Associated with anorexia/bulimia nervosa (self-induced vomiting), alcoholic cirrhosis, diabetes mellitus [2]
- Drug-induced (e.g., Phenytoin) [2]
- Sjögren's syndrome
Due to field cancerization, H&N cancer patients can develop second primaries. When a new lesion is found:
- Metastasis: Same histology, connected via lymphatic/haematogenous route
- Second primary (synchronous/metachronous): Different histology OR different location with dysplasia-carcinoma sequence, no intervening malignant tissue
- 10% risk of synchronous/metachronous tumour (field cancerization) [3] — this is why panendoscopy is mandatory
The lecture provides clear referral criteria [3]:
EARLY REFERRAL to ENT Surgeons when suspecting malignancy [3]:
- Persistent 2–4 weeks after conservative/empirical treatment [3]
- Clinically suspicious: irregular, induration, > 2cm, associated cervical LN enlargement [3]
Additional red flags from history and examination [2][3]:
- Age > 40 with smoking/alcohol history and new neck mass
- Hoarseness, dysphagia, otalgia in the context of a neck mass → suggests cervical lymph node metastasis from an underlying upper aerodigestive tract malignancy [2]
- Unilateral serous otitis media in an adult (think NPC)
- Progressive dysphagia with weight loss
- Blood-stained nasal discharge/saliva/sputum
- Non-healing oral ulcer > 3 weeks
- New-onset cranial nerve palsy
- 15–20% of oral cavity cancers have occult nodal metastasis — hence elective neck dissection is indicated even when the neck is clinically negative [3]
Oral cavity malignancy subsites [3]:
- Oral tongue (commonest)
- Buccal mucosa
- Floor of mouth
- Upper or lower alveolus
- Hard palate
- Lip
Clinical features of oral cavity SCC [3]:
- Exophytic mass
- Non-healing ulcer
- Painless at first, painful when infiltrating nerve
- Surrounding leukoplakia/erythroplakia
- Induration or fixation
- Loosened tooth +/- non-healing tooth socket
- Bleeding, swallowing/speech difficulty (ankyloglossia)
- 15–20% of occult nodal metastasis → Elective neck dissection
Ankyloglossia in Cancer Context
Ankyloglossia literally means "anchored tongue" (from Greek ankylos = crooked/fused, glossa = tongue). In the cancer context, this refers to restricted tongue mobility due to tumour infiltration into the floor of the mouth or extrinsic tongue muscles — the tongue becomes "tethered." Not to be confused with congenital tongue-tie.
| Presenting Complaint | Think First (Most Likely) | Must Not Miss | Important to Differentiate |
|---|---|---|---|
| Non-healing oral ulcer | SCC | Lymphoma, Syphilitic chancre | Traumatic ulcer, Aphthous |
| Asymmetric tonsil | SCC of tonsil | Lymphoma | Peritonsillar abscess (acute, febrile) |
| Neck mass (> 40, smoker) | Metastatic SCC | Lymphoma | Thyroid, Salivary gland tumour |
| Neck mass (young adult) | Branchial cleft cyst | Cystic metastasis from HPV+ SCC | Lymphoma, Thyroglossal cyst |
| Parotid mass | Pleomorphic adenoma | Malignant salivary tumour (if CN VII palsy) | Warthin's tumour, Intraparotid LN |
| Bilateral parotid swelling | Sialadenosis, Sjögren's | Lymphoma | Parotitis, Bruxism/masseter hypertrophy |
| Hoarseness > 3 weeks | Glottic SCC (if risk factors) | Laryngeal cancer, Lung apex tumour | Vocal cord polyp, Reinke's oedema, Reflux laryngitis |
| Unilateral serous otitis media (adult) | NPC | — | Eustachian tube dysfunction |
| Unilateral nasal obstruction + epistaxis | NPC, Sinonasal malignancy | Juvenile nasopharyngeal angiofibroma (adolescent male) | Nasal polyp, Inverted papilloma |
High Yield Summary
The Big Three DDx Categories: Infective (acute, febrile) vs Neoplastic (chronic, afebrile) vs Congenital/Developmental.
90% of H&N malignancies are SCC (excluding nasopharynx and thyroid).
Oral cavity/oropharynx histological DDx: SCC (epithelial, ulcerative) vs Lymphoma/minor salivary gland tumour (submucosal, smooth).
Key mimics of cancer: Lymphoma (tonsil/tongue base), syphilitic chancre (oral ulcer), branchial cleft cyst (cystic neck mass — may be HPV+ metastasis).
Red flags for malignancy referral: Persistent > 2–4 weeks, irregular, indurated, > 2cm, associated cervical LN. Hoarseness/dysphagia/otalgia with a neck mass suggests metastatic UADT cancer.
FNA limitations: Cannot provide tissue architecture → cannot subtype lymphoma. Need core/excisional biopsy.
Facial nerve palsy with parotid mass = malignant until proven otherwise.
15–20% occult nodal metastasis in oral cavity SCC → elective neck dissection.
Field cancerization → 10% risk synchronous/metachronous tumours → always panendoscopy.
Cystic Level II node in young adult: think HPV+ oropharyngeal SCC, not just branchial cleft cyst.
Active Recall - DDx of Head and Neck Cancer
References
[2] Senior notes: felixlai.md (H&N cancer, CA Oropharynx, NPC, Laryngeal carcinoma, Salivary gland, Neck mass sections) [3] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (pp. 34, 35, 36, 40, 41, 42, 48)
Diagnosis of Head and Neck Cancer
The diagnosis of H&N cancer follows a logical, stepwise approach built on first principles: find the lesion → prove it's cancer → determine how far it's spread → stage it → plan treatment. Every investigation you order should serve one of these purposes. Let's walk through this systematically.
H&N cancer does not have formal diagnostic criteria in the way that, say, rheumatoid arthritis has the ACR/EULAR criteria. Instead, the diagnosis is histopathological — you need a tissue biopsy showing malignant cells. Everything else (history, examination, imaging) serves to:
- Raise clinical suspicion (symptoms + risk factors + signs)
- Locate the primary tumour (endoscopy + imaging)
- Obtain tissue (biopsy or FNA)
- Stage the disease (TNM staging using imaging + pathology)
The "diagnostic criteria" for H&N cancer is therefore:
- Clinical suspicion (red flags, risk factors) → confirmed by histopathological demonstration of malignancy on biopsy
The Diagnosis is Histological
You cannot diagnose H&N cancer on imaging alone. A CT showing a nasopharyngeal mass is not a diagnosis of NPC — it is a finding that requires biopsy confirmation. The definitive diagnosis is ALWAYS tissue-based.
The workup follows a structured pathway. The lecture slides lay it out clearly [3]:
Workup and Investigation [3]:
- History
- Physical Examination
- Panendoscopy + biopsy — 10% risk of synchronous/metachronous tumour (field cancerization)
- Tonsillectomy or EUA + Bx
- Ultrasound neck +/- FNAC
- CXR
- CT/MRI
- PET scan if necessary
This was covered in Part 1 (Clinical Features), but in the diagnostic context the key points are:
- Age, Sex
- Duration: Acute vs Chronic — acute + febrile = infection; chronic + afebrile = neoplasm
- Symptoms — systematically cover ear, nose, mouth, throat, pharynx, neck (as per Part 1)
- Risk factors: smoking, alcohol, family history [3]
- Functional disturbances: breathing, chewing, swallowing, phonation, articulation [3]
- Co-morbidities — determines fitness for surgery/chemoradiation
Growth pattern of mass [2]:
- Present for years with minimal change → likely benign neoplasm
- Rapidly expanding → likely malignancy
Associated symptoms that suggest specific diagnoses [2]:
- Fever, night sweats, weight loss → suggests lymphoma
- Hoarseness, dysphagia, otalgia → suggests cervical lymph node metastasis from an underlying upper aerodigestive tract malignancy
4. Physical Examination
Physical Examination [3]:
Oral cavity and Oropharynx [3]:
- Systematic to all sub-sites
- Inspection and palpation (underlying mass/induration)
Why palpation? Because inspection alone misses submucosal disease. Bimanual palpation of the floor of mouth (one finger intraoral, one finger extraoral under the chin) can detect indurated masses that look normal on the surface. Palpation of the tongue base can reveal a mass invisible to the naked eye.
Neck [3]:
- Location (region/level)
- Shape + size (measure)
- Consistency
- Mobility
- Inflammation
Scalp/Skin [3] — don't forget: metastatic skin SCC/melanoma to parotid or cervical nodes is a recognised pathway. Always check the scalp and face.
Mass characterization features [2]:
- Rock-hard, fixed and non-tender → Malignancy
- Firm, rubbery, rapidly expanding → Lymphoma
- Discrete, mobile, firm, slightly tender → Reactive LN
- Pulsatile with bruit → Vascular lesion
Other examinations [7]:
- Complete H&N ENT examination
- Facial nerve examination — essential when salivary gland malignancy is suspected
- Palpation of neck lymph nodes
- Endoscopy of the upper aerodigestive tract
Especially important for NPC (skull base invasion → CN III–VI in cavernous sinus; parapharyngeal space → CN IX, X, XII, sympathetic chain). Also important for salivary gland tumours (CN VII for parotid).
This is the bedside scope — performed in the clinic with topical anaesthesia. It allows you to visualise the nasopharynx, oropharynx, hypopharynx, and larynx in a single pass.
Key findings:
- Nasopharyngeal mass (especially in Fossa of Rosenmüller) → NPC
- Asymmetric tonsil, tongue base mass → Oropharyngeal cancer or lymphoma
- Vocal cord mobility: Fixed cord = invasion of recurrent laryngeal nerve or cricoarytenoid joint fixation
- Pyriform sinus pooling → obstruction at hypopharynx/upper oesophagus
- Supraglottic/glottic/subglottic mass → laryngeal cancer
5. Investigation Modalities
5.1 Pathological Tests — The Core of Diagnosis
Fine needle aspiration — cytology [7][2]:
- Technique: 21–25 gauge needle, direct palpation or under ultrasound or CT guidance [2]
- Utility: Aspirate is used for cytological analysis, PCR testing for virus [2]:
- Limitation: Does NOT provide material for tissue architecture or immunohistochemical analysis [2]. This means FNA can tell you "these are malignant squamous cells" but cannot subtype a lymphoma (which requires assessment of tissue architecture and immunophenotyping).
- USG has limited use in oropharyngeal cancer but is a useful adjunct for FNAC to ensure accurate aspiration of a deeply seated lymph node swelling [2]
Why FNA first? Because it is minimally invasive, can be done in clinic, and gives rapid results. It is the first-line tissue sampling method for a neck mass.
- Obtains a cylinder of tissue → provides architecture as well as cells
- Useful when FNA is non-diagnostic or when lymphoma is suspected (architecture needed for subtyping)
- Risks: bleeding, nerve injury, tumour seeding (theoretical)
- Incisional biopsy should be performed in all cases of oropharyngeal/oral cavity suspicious lesions [2]
- Takes a wedge of tissue from the lesion edge (include normal and abnormal tissue)
- Gold standard for diagnosis of mucosal H&N cancers
Incisional vs Excisional Biopsy — A Critical Distinction
Excisional biopsy of a neck node is discouraged since it can adversely affect the success of subsequent surgical resection by field contamination in certain malignancy [2]. However, it is considered in the setting when FNA or core needle biopsy is positive for lymphoma where additional tissue is required for subtyping [2]. In short: for SCC — do NOT excise the node first (it can seed the surgical field and compromise the definitive neck dissection). For lymphoma — you may need an excisional biopsy because architecture is essential.
Tonsillectomy or EUA + Bx [3]
When the primary is occult (neck node metastasis but no visible primary), directed biopsies of likely primary sites are essential [7]:
Surgery in searching primary malignancy [7]:
- Right tonsillectomy & frozen section
- Left tonsillectomy
- Pharyngoscopy biopsy of hypopharynx & tongue base
- Nasopharyngoscopy & biopsy
Why tonsillectomy? Because HPV-positive oropharyngeal cancers can hide deep within the tonsillar crypts and be invisible on surface examination. A superficial biopsy may miss them. Ipsilateral tonsillectomy (on the side of the neck mass) followed by contralateral tonsillectomy if negative, with frozen section, is the standard approach for unknown primary workup.
| Test | Purpose | Subsite Relevance |
|---|---|---|
| p16 immunohistochemistry | Surrogate marker for HPV-driven tumour (p16 is overexpressed when Rb is inactivated by E7) | Oropharyngeal SCC |
| HPV DNA PCR / in situ hybridisation | Confirms HPV infection if p16+ | Oropharyngeal SCC |
| EBER in situ hybridisation | Detects EBV-encoded small RNA in tumour cells | NPC |
| Immunohistochemistry panel | CD20, CD3, Ki-67, etc. for lymphoma subtyping | Tonsillar/tongue base lymphoma |
| Test | Purpose | Interpretation |
|---|---|---|
| CBC with differentials [2] | Baseline; leukocytosis may suggest infection; pancytopenia may suggest marrow involvement | Non-specific |
| Serum ALP level [2] | ↑ ALP level in bone metastasis (from NPC or other H&N cancers with distant spread) | Elevated = investigate bones |
| Plasma EBV DNA level [2] | Detected by PCR. Diagnostic and staging evaluation for prognosis. Pre-treatment levels correlate with outcomes. Post-treatment levels evaluate treatment response and detection of recurrence | Quantitative — higher levels = larger tumour burden. Undetectable post-treatment = good response. Rising post-treatment = recurrence |
| EBV serology [2] | Sustained rise in IgA to both VCA and EA (↑ EBV VCA-IgA and EBV EA-IgA) | EBV-specific serological screening has low specificity for NPC [2]. Elevated titre may precede diagnosis of NPC by up to 10 years [2]. Used for population screening in endemic areas (HK). |
| Inflammatory markers (ESR, CRP) [2] | Rule out infection/inflammatory cause | Elevated in infection; also non-specifically elevated in malignancy |
| HIV serology [2] | Relevant if lymphoepithelial cyst, Kaposi's sarcoma, lymphoma suspected | |
| Autoantibodies (Anti-Ro/SSA, Anti-La/SSB) [2] | When Sjögren's disease is suspected as a cause of periparotid or submandibular masses |
EBV DNA vs EBV Serology — Know the Difference
Plasma EBV DNA (PCR) is quantitative, correlates with tumour burden, and is the preferred tool for monitoring treatment response and detecting recurrence in NPC. EBV serology (VCA-IgA, EA-IgA) is used for population-level screening in endemic areas but has low specificity (many healthy Southern Chinese have elevated EBV antibodies). In clinical practice for a known NPC patient, you follow the plasma EBV DNA, not the serology.
5.3 Imaging Studies
Ultrasound neck +/- FNAC [3]
- Non-invasive real-time assessment of mass and its relation to adjoining structures [2]
- Guide fine-needle aspiration [2]
- Best for: Evaluating cervical lymph nodes, thyroid nodules, salivary gland masses
- Limited for: Deep structures (skull base, parapharyngeal space, retropharyngeal nodes), mucosal primary tumours
Investigation of malignant salivary gland tumour — Bedside USG [7]:
- Tumour vs inflammation
- Location of tumour
- Cervical LN
Sonographic features of pathological lymph nodes [2]:
- Size > 1.0 cm in minimal axial diameter
- Rounded-shaped (loss of normal oval/kidney-bean shape)
- Increased or heterogeneous contrast enhancement
- Loss of normal fatty hilum (the echogenic centre disappears when replaced by tumour cells)
- Presence of central necrosis (hypoechoic/anechoic centre — tumour outgrows its blood supply)
Why these features? Normal lymph nodes are oval (long-axis/short-axis ratio > 2), have a preserved fatty hilum (echogenic centre), and are < 1cm short-axis. Metastatic nodes become rounded (tumour cells expand the cortex uniformly), lose the hilum (replaced by tumour), and develop necrosis centrally (tumour outgrows blood supply).
CT/MRI [3]
Strengths of CT [2]:
- CT is superior to MRI in terms of providing greater spatial resolution, faster acquisition time, and better for evaluation of bony destruction
- Determine extent of tumour infiltration into deep tongue musculature and whether mandible is involved for cancer of the oral cavity [2]
- Determine local invasion or infiltration into adjacent structures for other H&N cancers that is difficult to detect on physical examination [2]
- Detection of cervical lymph node metastasis [2]
- CT thorax and abdomen to assess for distant metastasis [2]
- Useful to detect bony invasion [2]
For salivary gland tumours — CT scan [7]:
- Bony invasion
- Cervical LN
CT features of pathological LNs [2] — same as USG but on cross-sectional imaging:
- Size > 1.0 cm in minimal axial diameter
- Rounded-shaped
- Increased or heterogeneous contrast enhancement
- Loss of normal fatty hilum
- Presence of central necrosis
When to choose CT over MRI: Bony detail needed (mandible invasion, skull base erosion pattern), fast acquisition (uncooperative patient, airway concerns), claustrophobic patient, CT chest/abdomen needed simultaneously for staging.
MRI scan [2]:
- MRI is superior to CT in terms of soft tissue delineation, detecting bone marrow invasion or skull base erosions
- Imaging modality of choice for cancer of the oral cavity and oropharynx [2] — because these sites require precise delineation of soft tissue extent (how far into the tongue muscles? is the pterygoid involved? is the parapharyngeal space invaded?)
- Provides optimal visualization of soft-tissue infiltration of the tumour [2]
- Detection of cervical lymph node metastasis [2]
For salivary gland tumours — MRI [7]:
- Accurate delineation of extent of invasion
- May be able to see nerve invasion (perineural spread along CN VII in parotid cancer — T2-weighted and post-gadolinium images show nerve thickening/enhancement)
For NPC: CT or MRI of nasopharynx, skull base and neck — Assessment of locoregional disease extent [2]
MRI indications for neck masses [2]:
- Infiltrative soft tissue masses
- Suspicious malignant perineural spread
- Potential CNS origin
When to choose MRI over CT: Soft tissue detail needed (tongue, palate, parapharyngeal space), skull base invasion assessment, perineural spread, no radiation exposure (younger patients).
| Feature | CT | MRI |
|---|---|---|
| Bone detail | Superior (cortical destruction) | Better for marrow invasion |
| Soft tissue | Adequate | Superior |
| Speed | Fast (seconds) | Slow (30–60 min) |
| Availability | More available | Less available |
| Perineural spread | Limited | Superior |
| Skull base | Good for bony erosion | Superior for soft tissue/cavernous sinus |
| Preferred for | Larynx (cartilage), chest/abdomen staging, bony invasion | Oral cavity, oropharynx, nasopharynx, salivary glands |
Panendoscopy + biopsy [3] — 10% risk of synchronous/metachronous tumour (field cancerization) [3]
Panendoscopy = Direct laryngoscopy + Bronchoscopy + OGD [2]
This is performed under general anaesthesia. It serves three purposes:
- Detection of synchronous primary cancer [2] — because of field cancerization, you must examine the entire UADT, tracheobronchial tree, and upper oesophagus.
- Part of operative staging for characterization of primary tumour [2] — assess tumour extent directly.
- Panendoscopy may identify synchronous primaries that are too small to be identified with PET scan [2] — a subtle mucosal lesion in the oesophagus visible on OGD with NBI may be missed on PET.
Key areas assessed during panendoscopy for laryngeal cancer [2]:
- Supraglottic tumour: Vallecula, base of tongue, ventricle, arytenoid, anterior commissure
- Glottic tumours: False cord, arytenoid, anterior commissure, subglottic extension
PET scan if necessary [3]
PET-CT scan [2]:
- Uses 18F-fluorodeoxyglucose (FDG) as contrast — cancer cells have high metabolic activity and take up more glucose (Warburg effect), so they "light up" on PET.
- PET scan is superior to both CT and MRI for detecting regional nodal metastasis as well as distant metastasis and second primary tumours [2]
- PET scan may identify lower aerodigestive tract tumours not seen with panendoscopy [2]
- Identify primary disease or detect distant metastatic disease [2]
For salivary gland tumours — PET-CT [7]:
- Workup for distant metastasis
For NPC [2]:
- PET-CT scan — Use of 18-fluorodeoxyglucose as contrast
- Superior ability to detect lymph node and bone metastasis
Indications for PET-CT in H&N cancer:
- Unknown primary — neck node SCC, primary not found on examination + CT/MRI → PET-CT may reveal the primary
- Staging of advanced disease — looking for distant metastases
- Post-treatment surveillance — detecting recurrence (especially differentiating post-radiation changes from residual/recurrent tumour)
- Assessment of treatment response — serial PET-CTs
Limitations: Poor spatial resolution compared to CT/MRI (can miss lesions < 1cm); false positives from inflammation/infection; physiological FDG uptake in Waldeyer's ring and muscles (can confuse interpretation).
| Modality | Indication |
|---|---|
| CXR [3][2] | Routine staging — evaluate for lung metastasis. Quick, cheap, baseline. |
| Bone scan [2] | Evaluate for bone metastasis — especially NPC (bone is most common distant metastasis site at 75%) |
| X-ray (Panoramic/OPG) [2] | Detects gross cortical invasion of mandible in oral cavity cancer |
| Angiography [7] | Vascular lesions (carotid body tumour) — preoperative planning, embolisation |
6. Staging — TNM System
Once the diagnosis is confirmed and workup complete, the tumour is staged using the AJCC 8th Edition TNM system (2017) [2].
| N Stage | Description |
|---|---|
| NX | Regional LN cannot be assessed |
| N0 | No regional LN metastasis |
| N1 | Metastasis in single ipsilateral LN < 3 cm and ENE− |
| N2a | Metastasis in single ipsilateral LN < 3 cm and ENE+ OR single ipsilateral 3–6 cm and ENE− |
| N2b | Metastasis in multiple ipsilateral LN, all < 6 cm and ENE− |
| N2c | Metastasis in bilateral or contralateral LN, all < 6 cm and ENE− |
| N3a | Metastasis in LN > 6 cm and ENE− |
| N3b | Metastasis in any node with ENE+ (except N2a criteria) |
ENE = Extranodal extension (previously called extracapsular spread). This is when tumour breaks through the lymph node capsule into surrounding soft tissue. It is a critical prognostic factor and upstages the N category [2].
Why does ENE matter? Because a tumour confined within a node capsule has a better prognosis than one that has burst through — the latter indicates more aggressive biology and higher risk of locoregional recurrence and distant metastasis.
| M Stage | Description |
|---|---|
| M0 | No distant metastasis |
| M1 | Distant metastasis |
NPC has its own N staging because of its unique lymphatic drainage pattern (bilateral, retropharyngeal nodes common, supraclavicular involvement has distinct prognostic significance).
AJCC 8th edition introduced separate staging for p16-positive oropharyngeal cancer. The N staging is simplified (N1 = unilateral nodes ≤ 6 cm; N2 = contralateral or bilateral nodes ≤ 6 cm; N3 = nodes > 6 cm) and overall stages are "downstaged" compared to HPV-negative tumours — reflecting the dramatically better prognosis.
| Subsite | Key Investigations | Key Findings |
|---|---|---|
| Oral cavity | Inspection + palpation + incisional biopsy; OPG for mandible; CT (bony invasion); MRI (imaging modality of choice) [2] | SCC histology; perineural invasion; DOI (depth of invasion, critical for T staging in AJCC 8th ed) |
| Oropharynx | Panendoscopy + biopsy; USG neck +/- FNAC; CT/MRI; p16 IHC and HPV testing [3][2] | p16+ = HPV-driven (better prognosis, separate staging) |
| NPC | Nasopharyngoscopy + biopsy (definitive diagnosis) [2]; Plasma EBV DNA (PCR) [2]; EBV serology (VCA-IgA, EA-IgA) [2]; CT/MRI nasopharynx, skull base, neck [2]; PET-CT [2]; CXR [2]; Bone scan [2] | EBER+ on ISH; Non-keratinizing undifferentiated carcinoma (most common in HK); Incisional nodal biopsy or nodal dissection should be AVOIDED since it will negatively impact subsequent treatment [2] |
| Hypopharynx | Panendoscopy + biopsy; CT/MRI; Barium swallow (occasionally, for postcricoid lesions) | Loss of laryngeal crepitus on exam |
| Larynx | Laryngoscopy (rigid/flexible fibreoptic) — allows biopsy to confirm histology, determines extent of local spread [2]; Panendoscopy (Direct laryngoscopy + Bronchoscopy + OGD) — exclude synchronous lesion [2]; CT/MRI neck — evaluate extent of tumour invasion, extra-laryngeal spread, suspicious LN involvement, cartilage erosion or invasion, extension into pre-epiglottic or paraglottic spaces [2] | Vocal cord fixation; cartilage invasion; paraglottic/pre-epiglottic space involvement |
| Salivary gland | FNA cytology or core needle biopsy [2]; USG (tumour vs inflammation, location, cervical LN) [7]; CT (bony invasion, cervical LN) [7]; MRI (accurate delineation of extent of invasion, nerve invasion) [7]; PET-CT (workup for distant metastasis) [7]; Facial nerve examination [7] | FNA can usually discriminate benign from malignant but is less specific for exact tumour type [2]; facial nerve palsy = malignant |
NPC Diagnostic Pearl
Incisional nodal biopsy or nodal dissection should be AVOIDED in NPC since it will negatively impact subsequent treatment [2]. NPC is treated primarily with radiotherapy ± chemotherapy, NOT surgery. An open neck dissection before radiation disrupts tissue planes and worsens outcomes. The diagnosis should be made from a nasopharyngoscopy-guided biopsy of the primary tumour, not from the neck node.
When a patient presents with a metastatic neck node (SCC on FNA) but no clinically visible primary tumour, this triggers the "unknown primary" workup. This is a common exam scenario.
Step-by-step approach [7][2][3]:
- FNA of neck node — confirms metastatic SCC. Send for HPV (p16) and EBV testing [2]:
- p16+ → primary likely in oropharynx (tonsil or tongue base)
- EBV+ → primary likely in nasopharynx
- Clinical examination — thorough H&N ENT examination including flexible nasopharyngolaryngoscopy
- CT/MRI — may reveal a subtle primary
- PET-CT — PET scan may identify lower aerodigestive tract tumours not seen with panendoscopy [2]; superior to CT and MRI for detecting regional nodal metastasis, distant metastasis, and second primary tumours [2]
- Surgery in searching primary malignancy [7]:
- Right tonsillectomy & frozen section
- Left tonsillectomy
- Pharyngoscopy biopsy of hypopharynx & tongue base
- Nasopharyngoscopy & biopsy
If the primary is still not found after all of this, treatment is directed at the neck (neck dissection ± radiation to likely mucosal sites).
| Investigation | What It Tells You | When to Use |
|---|---|---|
| FNA/FNAC | Cytology, HPV/EBV PCR | First-line for neck mass; adjunct for deeply seated nodes |
| Core needle biopsy | Architecture + IHC | Suspected lymphoma; FNA non-diagnostic |
| Incisional biopsy | Definitive histology of primary | All oral/oropharyngeal mucosal lesions |
| Tonsillectomy / EUA + Bx | Occult primary search | Unknown primary workup |
| USG neck | Node assessment, guide FNA | All neck masses |
| CT | Bony invasion, nodal disease, chest/abdomen staging | All H&N cancers |
| MRI | Soft tissue extent, perineural spread, skull base | Oral cavity, oropharynx, NPC, salivary glands |
| Panendoscopy | Synchronous tumours, tumour extent | All UADT cancers (field cancerization — 10% risk) |
| PET-CT | Distant metastasis, unknown primary, recurrence | Advanced disease, unknown primary, post-treatment |
| CXR | Lung metastasis, second primary lung cancer | Baseline for all |
| Bone scan | Bone metastasis | NPC, symptomatic patients, elevated ALP |
| Plasma EBV DNA | Tumour burden, treatment response, recurrence | NPC (diagnostic, monitoring) |
| EBV serology (VCA-IgA) | Screening in endemic populations | Population screening; low specificity for individual diagnosis |
| p16 IHC + HPV PCR | HPV-driven oropharyngeal cancer identification | All oropharyngeal SCC |
High Yield Summary
Diagnosis is histological — always need tissue (biopsy/FNA). Cannot diagnose on imaging alone.
Workup sequence: History + PE → Endoscopy + Biopsy → USG + FNAC → CT/MRI → PET-CT if necessary → TNM staging → MDT.
FNA: First-line for neck mass. Can do cytology + HPV/EBV PCR. Cannot subtype lymphoma (no architecture).
Panendoscopy (laryngoscopy + bronchoscopy + OGD): Mandatory for all UADT cancers — 10% synchronous/metachronous tumour risk from field cancerization.
CT vs MRI: CT for bone and speed; MRI for soft tissue, perineural spread, skull base. MRI is imaging of choice for oral cavity/oropharynx; CT crucial for larynx (cartilage invasion).
PET-CT: Superior to CT/MRI for nodal metastasis, distant metastasis, and second primaries. Used for unknown primary and post-treatment surveillance.
NPC workup: Nasopharyngoscopy-guided biopsy (NOT nodal biopsy/dissection); Plasma EBV DNA for monitoring; EBV serology for screening (low specificity).
TNM staging: T = site-specific; N = uniform (except NPC); M = uniform. ENE upstages N category. HPV+ oropharyngeal cancer has separate staging (AJCC 8th ed).
Unknown primary: FNA → p16/EBV testing → PET-CT → Panendoscopy + directed biopsies + bilateral tonsillectomy.
Sonographic features of malignant LN: Size > 1cm short-axis, rounded, loss of fatty hilum, central necrosis, heterogeneous enhancement.
Active Recall - Diagnosis of Head and Neck Cancer
References
[2] Senior notes: felixlai.md (H&N cancer diagnosis, TNM staging, NPC, CA Oropharynx, Laryngeal carcinoma, Neck mass, Salivary gland sections) [3] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (pp. 9, 35, 36, 40, 41, 42) [7] Lecture slides: GC 218. I have a swelling in the neck Neck mass.pdf (pp. 7, 9); GC 217. Facial nerve palsy and salivary gland diseases.pdf (pp. 42, 77)
Management of Head and Neck Cancer
Management of H&N cancer is one of the most complex areas in oncology because you are constantly balancing two competing goals: cure the cancer and preserve the patient's ability to breathe, eat, speak, and look normal. No other cancer site imposes this dual burden so acutely.
Let's build the management framework from first principles before diving into subsite-specific details.
1. Overarching Management Principles
General principle [3]:
- Tumour clearance with long-term survival benefit
- Organ and function preservation
When surgery is indicated [3]:
- Resection with adequate margins
- Reconstruction for Form and Function
- Rehabilitation always — swallowing, voice and hearing
Think of this as the "3 R's" of H&N cancer surgery: Resection → Reconstruction → Rehabilitation.
Therefore, majority of head and neck cancers require both RESECTION and RECONSTRUCTION [5]
Aims of Head and Neck Surgery [5]:
- Resection: oncologically clear, yet preserve important organ functions → margins of resection
- Reconstruction: choose the best option for individual patient
Why both? Because H&N structures are not expendable. If you resect a colon tumour, the patient can live without that segment. If you resect the tongue, the patient cannot eat or speak without reconstruction. The surgery must simultaneously achieve oncological clearance AND restore function.
Head and neck cancer poses special challenges in both resection and reconstruction. Individualise the option of surgery to achieve the best functional and cosmetic result [5]
Multidisciplinary approach including swallowing rehabilitation is important [2]
A H&N cancer MDT typically includes:
- Head and neck surgeon (ENT / maxillofacial / plastic surgery)
- Radiation oncologist
- Medical oncologist
- Pathologist
- Radiologist
- Speech and language therapist
- Dietitian
- Clinical psychologist / psychiatrist
- Dental specialist (pre-radiation dental assessment — critical to prevent osteoradionecrosis)
- Nurse specialist
ALWAYS protect the airway for all H&N cancer [2]. Before any treatment decision, assess the airway. A patient with impending airway obstruction needs urgent intervention (tracheostomy / intubation) before any staging or treatment planning.
The lecture gives us the overarching framework clearly [3]:
Management Framework — Based on TNM staging [3]:
Early stage (I, II) [3]:
- Single modality of treatment
- Surgery or radiotherapy alone
Late stage (III, IV) [3]:
- Combined modality of treatment
- Concurrent chemo-irradiation
- Surgery with adjuvant radiotherapy +/- chemotherapy
General rule [3]:
- Early stage: radiotherapy or minimally invasive surgery (laser/robotic)
- Late stage: Surgery with adjuvant treatment
BUT [3]:
- Oral cavity and thyroid: surgery in early stage (Why? Because oral cavity cancers are not as radiosensitive as pharyngeal/laryngeal cancers, and surgical access is straightforward)
- NPC: chemo-irradiation in late stage (Why? Because NPC is highly radiosensitive — especially the undifferentiated, EBV-associated subtype endemic in HK — and the nasopharynx is anatomically difficult to access surgically, sitting deep at the skull base)
The Key Exception Rule
The general rule is "early = single modality, late = combined modality." But the two big exceptions are: (1) Oral cavity → surgery first even in early stage (not as radiosensitive; easy surgical access); (2) NPC → chemo-irradiation even in advanced stage (highly radiosensitive; surgically inaccessible). If you remember nothing else about H&N management, remember this.
4. Treatment Modalities in Detail
4.1 Surgery
The surgical approach depends on subsite, tumour size, and accessibility.
Access approaches:
- Traditional surgical approaches through skin incision — used for salivary and thyroid cancers which are easily accessible [2]
- Minimally invasive techniques: [2]
- Transoral laser microsurgery (TOLM) — for larynx cancers
- Transoral robotic surgery (TORS) — for oropharynx cancers
- Previously, procedures might otherwise require a lip-splitting mandibulotomy approach to resect tumours of the oropharynx [2]. TORS avoids this.
- TORS is associated with shorter length of hospital stay and less likely to be gastrostomy tube or tracheostomy dependent at 6 months [2]
Margin adequacy:
- The goal is R0 resection (negative margins). For oral cavity SCC, AJCC 8th edition recommends ≥ 5mm clear margin. Intraoperative frozen section helps guide real-time margin assessment.
- Postoperative RT ± chemotherapy is indicated for patients who have close or positive margins or factors increasing risk of local recurrence including lymphovascular invasion, perineural invasion, and extranodal extension (ENE) [2]
Subsite-specific surgery — Oral Cavity:
| Subsite | Early Disease | Advanced Disease |
|---|---|---|
| Lip | Surgical excision with histological confirmation of tumour-free margin [2]. Wedge excision with primary closure for small lesions. | Larger resection + reconstruction |
| Tongue | Wide local excision with primary closure or healing by secondary intention (T1–2) [2]. CO2 ablation for premalignant lesions [2] | Partial glossectomy + Reconstruction using soft pliable fasciocutaneous free flaps [2]. Prosthetic augmentation can allow contact between remaining tongue tissue and palate, improving ability to speak and swallow [2] |
| Floor of mouth | Wide local excision + split-thickness skin graft [2] | Marginal or segmental mandibulectomy + Reconstruction with fasciocutaneous or vascularized osseous free flap [2] |
| Alveolus/Gingiva | Marginal resection of mandible for minimal bone invasion [2] | Segmental mandibulectomy for invasion into medullary cavity [2] |
| Retromolar trigone | — | Marginal or segmental mandibulectomy with soft tissue or osseous reconstruction [2]. Ipsilateral neck dissection [2] |
| Buccal mucosa | Surgical excision [2] | Combined surgical excision + post-op RT. May necessitate resection of alveolar ridge [2] |
| Hard palate | Mucosal excision (periosteum acts as barrier) [2] | Partial palatectomy or infrastructure maxillectomy. Through-and-through defects require dental prosthesis [2] |
Subsite-specific surgery — Larynx:
Early (Stage I and II) cancer [2]:
- Patients with early cancer should be treated with the intent to preserve the larynx
- Larynx-preserving surgery: [2]
- Transoral laser microsurgery (TOLM)
- Partial open laryngectomy
Advanced (Stage III and IV) cancer [2]:
- Patients with good performance status should be treated with a functional organ-reservation strategy including chemoradiotherapy or larynx-preserving surgery with post-operative RT
- Total laryngectomy is indicated for: [2]
- Patients who are not candidates for functional organ-reservation strategy
- Elderly patients or patients with poor functional status that cannot tolerate the associated toxicities of chemoradiotherapy
- Patients with resectable tumours with destruction of both vocal cords or extensive cartilage destruction such that vocal or airway protective function cannot be recovered
- Total laryngectomy + Post-operative radiotherapy [2]
- Reconstruction by means of a pectoralis major flap or free flap reconstruction is required for lesions with pharyngeal extension [2]
Treatment of subglottic tumours [2]:
- Subglottic tumours are the least common and are commonly asymptomatic until locally advanced
- Higher rates of local recurrence and poor survival when compared with lesions involving the supraglottis or glottis
- Treatment is aggressive with initial surgical treatment consisting of either total laryngectomy or partial laryngectomy in very selected cases
- Thyroidectomy and bilateral paratracheal node dissection are usually performed — because the subglottis drains to paratracheal and pretracheal nodes, and the thyroid gland is in direct anatomical continuity
The neck must be addressed because of the high rate of regional metastasis. There are three types:
| Type | What is Removed | Indication |
|---|---|---|
| Selective neck dissection | Selected levels at highest risk of metastasis (e.g., Levels I–III for oral cavity; Levels II–IV for oropharynx/larynx) | Indicated for patients with stage I oral tongue cancer > 3mm and most stage II oral cavity cancers [2]. Also for clinically N0 necks with > 15–20% risk of occult metastasis. Ipsilateral selective neck dissection of level I to III or IV is generally sufficient [2]. Bilateral neck dissection required if primary tumours are close to or involving the midline [2] |
| Modified radical neck dissection | Levels I–V LN. Preservation of one or more of: internal jugular vein, spinal accessory nerve, sternocleidomastoid muscle [2] | Clinically positive neck (palpable/imaged nodes) |
| Radical neck dissection | Levels I–V LN + Internal jugular vein + Spinal accessory nerve + Sternocleidomastoid muscle [2] | Rarely performed now; reserved for bulky nodal disease with gross invasion of these structures |
Why preserve the spinal accessory nerve (CN XI)? Because its sacrifice causes trapezius muscle denervation → shoulder drop, chronic pain, inability to abduct arm above 90°. This is devastating for quality of life. Modern practice tries to spare it whenever oncologically safe.
Majority of head and neck cancers require both RESECTION and RECONSTRUCTION [5]
| Reconstructive Option | When Used |
|---|---|
| Primary closure | Small defects (e.g., small tongue excision, lip wedge excision) |
| Split-thickness skin graft | Floor of mouth defects after excision of superficial lesions |
| Local flaps | Lip reconstruction (e.g., Abbe flap, Estlander flap), small oral defects |
| Pedicled flaps (e.g., pectoralis major myocutaneous flap) | Moderate to large defects; reliable blood supply; no need for microvascular anastomosis |
| Free flaps (fasciocutaneous, e.g., radial forearm; osseous, e.g., fibula free flap) | Reconstruction using soft pliable fasciocutaneous free flaps can provide intraoral bulk and preservation of tongue mobility [2]. Fibula free flap for mandibular reconstruction. Gold standard for complex defects |
| Dental prosthesis | Through-and-through defects of palate require dental prosthesis for rehabilitation of swallowing and speech [2]. Obturators for palatal defects |
| Prosthetic augmentation | Can allow contact between remaining tongue tissue and palate, improving ability to speak and swallow [2] |
4.2 Radiotherapy (RT)
"Radio" = radiation; "therapy" = treatment. Uses ionising radiation to cause DNA double-strand breaks in tumour cells → cell death.
| Type | Description | Use |
|---|---|---|
| External beam RT (EBRT) | Radiation delivered from outside the body. Modern techniques include intensity-modulated RT (IMRT) and volumetric modulated arc therapy (VMAT) — these shape the radiation beam to conform to the tumour and spare surrounding normal tissue | Standard RT for H&N cancer |
| Brachytherapy | Radioactive source placed directly into or adjacent to the tumour | Used for localised early-stage cancers [2]; interstitial brachytherapy for lip, oral tongue, floor of mouth |
| Proton beam therapy | Uses protons instead of photons; deposits energy more precisely (Bragg peak) → less exit dose | Emerging role for NPC (to spare temporal lobes and brainstem) and skull base tumours |
| Setting | Indication |
|---|---|
| Definitive RT (single modality) | Early-stage laryngeal cancer (equally effective as surgery with better voice quality) [2]; NPC Stage I (RT only) [2]; Early oropharyngeal cancer |
| Post-operative RT (adjuvant) | Indicated for patients with close or positive margins, factors increasing risk of local recurrence: lymphovascular invasion, perineural invasion, ENE [2]; Post-op RT for lip cancer: positive margins, tumour thickness > 4mm, perineural invasion, LN metastasis [2]; Post-op RT for hard palate: advanced staged tumours [2] |
| Concurrent chemoradiotherapy | Advanced stage (III and IV) oropharyngeal carcinoma — effectively preserves function and associated with survival comparable to surgery with post-operative radiation [2]; NPC Stage II–IVB [2] |
Radiotherapy is often preferred for early glottic cancer because of better functional outcomes, particularly voice quality, while avoiding GA and other risks associated with surgery [2]
Radiation therapy is equally as effective as surgery in controlling disease for early-stage cancers of the glottis [2]
Complications of RT for laryngeal cancer [2]: Radiation dermatitis, Hoarseness, Dysphagia, Odynophagia
Why RT for Early Glottic but Surgery for Early Oral Cavity?
Glottic cancer: The true vocal cord is exquisitely sensitive to even small changes in mass or stiffness. Surgery on the vocal cord (even laser excision) can cause scarring that permanently impairs voice quality. RT achieves similar cure rates but with better voice outcomes. That's why RT is often preferred.
Oral cavity cancer: The oral cavity mucosa is less radiosensitive than pharyngeal/laryngeal mucosa. RT to the oral cavity causes significant mucositis, xerostomia, and risk of osteoradionecrosis of the mandible. Surgery is straightforward (direct access through the mouth) and gives excellent local control. So surgery is preferred upfront.
Chemotherapy in H&N cancer is almost never used as a standalone curative modality. It is used as:
| Setting | Agents | Purpose |
|---|---|---|
| Concurrent chemoradiotherapy | Cisplatin (standard; 100 mg/m² q3 weeks × 3 cycles during RT) | Radiosensitiser — cisplatin inhibits DNA repair in tumour cells, making them more susceptible to radiation-induced damage. The combination is synergistic. |
| Induction (neoadjuvant) chemotherapy | TPF regimen: Docetaxel + Cisplatin + 5-Fluorouracil | Shrink bulky tumours before definitive treatment (RT or surgery). Used in organ preservation protocols for larynx and selected oropharynx/hypopharynx cancers. |
| Adjuvant chemotherapy | Cisplatin-based | After concurrent CRT for NPC (especially Stage III–IVB); reduces distant metastasis risk [2] |
| Palliative chemotherapy | Platinum-based combinations, taxanes | Incurable metastatic/recurrent disease — intent is symptom control and life prolongation, not cure |
Why cisplatin? Cisplatin ("cis-diaminedichloroplatinum") is a platinum-based drug that forms DNA cross-links → blocks DNA replication and transcription → triggers apoptosis. It is the most effective single agent in HNSCC and the backbone of concurrent CRT protocols.
| Agent | Target | Indication |
|---|---|---|
| Cetuximab | EGFR (Epidermal Growth Factor Receptor) | Alternative to cisplatin in concurrent CRT for patients who cannot tolerate cisplatin (e.g., renal impairment, elderly). "Cetuxi-mab" = chimeric monoclonal antibody against EGFR. EGFR is overexpressed in ~90% of HNSCC. Cetuximab blocks ligand binding → inhibits downstream Ras/MAPK and PI3K/Akt proliferation signals. |
| Agent | Target | Indication |
|---|---|---|
| Pembrolizumab | PD-1 (Programmed Death-1 receptor) | First-line for recurrent/metastatic HNSCC (KEYNOTE-048 trial). "Pembro-lizumab" = humanised monoclonal antibody against PD-1. PD-1 is an immune checkpoint receptor on T cells. Tumour cells express PD-L1 which binds PD-1 → switches off T-cell attack. Pembrolizumab blocks this interaction → T cells can recognise and kill tumour cells again. |
| Nivolumab | PD-1 | Second-line for platinum-refractory recurrent/metastatic HNSCC (CheckMate-141 trial). |
Immunotherapy has transformed the landscape for recurrent/metastatic HNSCC. In the curative setting, multiple trials are ongoing combining immunotherapy with CRT.
5. Subsite-Specific Management
Oral cavity: surgery in early stage [3]
Early (Stage I–II) [2]:
- Primary surgery is recommended rather than definitive radiotherapy
- Selective neck dissection indicated for stage I oral tongue cancer > 3mm depth of invasion and most stage II oral cavity cancers
- Post-operative radiotherapy if tumour thickness > 4mm or lymphovascular/perineural invasion
Advanced (Stage III–IV) [2]:
- Surgical resection is suggested as initial therapy
- Modified radical neck dissection + Post-operative radiotherapy ± concurrent chemotherapy
- Definitive radiotherapy or chemotherapy for medically inoperable or unresectable disease
Tumours of the oropharynx tend to be chemosensitive [2] Adequate treatment of the neck is important because of high risk of regional metastasis [2]
Early (Stage I–II): Single modality — surgery (TORS) or RT alone [3]
Advanced (Stage III–IV) [2]:
- Concomitant chemoradiation is commonly utilised in advanced stage (III and IV) oropharyngeal carcinoma, which effectively preserves function and is associated with survival comparable to surgery with post-operative radiation
- Alternatively: Surgery + post-op CRT
HPV-positive oropharyngeal cancer [2]:
- De-intensification of treatment can be considered while obtaining the same locoregional and overall survival seen with standard treatment options
- Active clinical trials are testing reduced-dose RT, replacing cisplatin with cetuximab, or reducing adjuvant treatment in good-responders
- The goal is to maintain the excellent survival while reducing long-term toxicity (xerostomia, dysphagia, fibrosis)
NPC: chemo-irradiation in late stage [3]
Management according to stages [2]:
| Stage | Approach |
|---|---|
| Early Stage I | Radiotherapy ONLY |
| Intermediate Stage II | Concurrent chemoradiotherapy |
| Advanced Stage III | Concurrent chemoradiotherapy |
| Stage IVA | ± Adjuvant chemotherapy |
| Stage IVB | ± Adjuvant chemotherapy |
Why RT-based for NPC? Three reasons:
- Anatomical: The nasopharynx sits deep at the skull base, surrounded by critical structures (carotid arteries, cranial nerves, brainstem). Surgical access is extremely difficult and morbid.
- Radiosensitivity: The undifferentiated (non-keratinizing) NPC — which is the dominant histology in Hong Kong — is highly radiosensitive.
- EBV-driven immune biology: These tumours have an immunogenic profile that responds well to CRT.
Standard protocol: Cisplatin 100 mg/m² q3 weeks × 3 during RT (concurrent CRT). For Stage III–IVB, adjuvant chemotherapy (gemcitabine + cisplatin) may follow based on the NPC-0501 and KEYNOTE-122 data.
Post-treatment follow-up [2]: Plasma EBV DNA monitoring — undetectable post-treatment = good response; rising levels = recurrence.
Early (Stage I and II) [2]:
- Radiotherapy is often preferred because of better functional outcomes, particularly voice quality
- Radiation therapy is equally effective as surgery in controlling disease for early-stage cancers of the glottis
- Larynx-preserving surgery: TOLM, partial open laryngectomy
Advanced (Stage III and IV) [2]:
- Functional organ-reservation strategy:
- Chemoradiotherapy (cisplatin + RT)
- Larynx-preserving surgery + post-op RT
- Total laryngectomy + post-op RT for:
- Cannot tolerate CRT toxicities
- Destruction of both vocal cords or extensive cartilage destruction
- Voice rehabilitation after total laryngectomy (tracheoesophageal puncture with voice prosthesis, oesophageal speech, or electrolarynx)
Surgery first for all stages [3] — because salivary gland cancers are generally not very radiosensitive (except lymphoepithelial carcinoma, which behaves like NPC).
Specific salivary malignancies (from lecture):
Mucoepidermoid Carcinoma [7]:
- Treatment influenced by site, stage, grade
- Localised to gland → Excision of gland
- Neck node metastasis → Neck dissection
- RT for high-grade tumour, close margin, and extra-glandular spread
Adenoid Cystic Carcinoma [7]:
- Complete local excision
- Tendency for perineural invasion — ? facial nerve sacrifice (this is one of the most agonising surgical decisions — do you sacrifice CN VII to get clear margins in a tumour known for perineural spread? The decision depends on whether the nerve is grossly involved vs. only microscopic perineural invasion on frozen section)
- Postoperative XRT
- Prognosis: Local recurrence 40%. Distant metastasis common: lung. Indolent course: 5-year survival 75%, 20-year survival 13%
Carcinoma Ex-Pleomorphic Adenoma [7]:
- Radical excision
- Neck dissection (25% with lymph node involvement at presentation)
- Postoperative XRT
- Prognosis: Dependent upon stage and histology. Usually not very good
Undifferentiated carcinoma — Lymphoepithelial carcinoma [7]:
- Histology exactly like NPC
- EBV-related (tumour cells EBER+ve)
- May present as metastatic lymph node, unknown primary
- Surgery + post-op RT to primary and neck
- Prognosis better than SCC and adenocarcinoma
Adenoid Cystic Carcinoma — The Long Game
ACC has an indolent but relentless natural history. Patients may do well for years, but the 20-year survival is only 13%. It is notorious for perineural invasion (spreading along nerves like a vine climbing a trellis) and late lung metastases (appearing 10–15 years after initial treatment). Long-term follow-up is essential.
Not every surgically-treated patient needs adjuvant therapy. It is indicated when adverse pathological features are present that increase the risk of locoregional recurrence [2]:
| Feature | Why It Matters | RT Alone Sufficient? |
|---|---|---|
| Close margin (< 5mm) | Residual microscopic disease at edges | Yes — RT to primary site |
| Positive margin (tumour at ink) | Very high recurrence risk | Consider re-excision first; if not feasible, CRT |
| Tumour thickness > 4mm | Deeper tumours have higher risk of occult nodal metastasis and local recurrence [2] | Yes — RT to primary + neck |
| Perineural invasion | Tumour cells spread along nerve sheaths → can recur far from the original resection site [2] | CRT (positive margin + PNI is particularly high risk) |
| Lymphovascular invasion | Increased risk of regional and distant spread [2] | RT |
| Extranodal extension (ENE) | Tumour through node capsule → dramatically increased recurrence and distant metastasis risk [2] | CRT (ENE + positive margin = the two strongest indications for adjuvant CRT based on EORTC 22931 and RTOG 9501 trials) |
| Multiple positive nodes | Higher nodal burden = higher risk | RT or CRT |
| Advanced T stage (pT3–4) | Larger tumours = higher local recurrence risk | RT |
The Two 'Absolute' Indications for Adjuvant Chemo-RT
The two features that mandate concurrent chemoradiotherapy (not just RT alone) after surgery are: (1) Positive margins and (2) Extranodal extension (ENE). These are the findings from EORTC 22931 and RTOG 9501 landmark trials. Other adverse features (PNI, LVI, close margins, multiple nodes, pT3–4) are indications for adjuvant RT, but the addition of chemotherapy is strongest for positive margins and ENE.
7. Contraindications and Special Considerations
| Contraindication | Rationale |
|---|---|
| NPC (relative) | Anatomically inaccessible; highly radiosensitive; surgery reserved for salvage recurrence |
| Medically unfit | Cardiac, pulmonary, or other comorbidities that preclude GA. ASA III–IV may be offered RT instead |
| Unresectable disease | Encasement of carotid artery, skull base invasion through to brain, prevertebral fascia invasion with fixation to spine |
| Distant metastatic disease (M1) | Surgery of primary is usually not curative in setting of widespread metastases (palliative intent only) |
| Contraindication | Alternative |
|---|---|
| Renal impairment (GFR < 50) | Carboplatin or cetuximab |
| Severe hearing loss | Carboplatin (cisplatin is ototoxic) |
| Severe neuropathy | Carboplatin |
| Poor performance status (ECOG ≥ 3) | RT alone or best supportive care |
Mandatory before starting RT to the H&N region. Why? Radiation damages salivary glands → xerostomia (dry mouth) → loss of protective saliva → rapid dental caries → tooth extraction in irradiated bone → osteoradionecrosis (ORN) of the mandible (a devastating complication). All carious teeth in the radiation field should be extracted and the sockets allowed to heal BEFORE starting RT (usually 2–3 weeks).
For patients with incurable metastatic/recurrent disease:
- Systemic therapy: Pembrolizumab ± chemotherapy (KEYNOTE-048); Cetuximab + platinum + 5-FU (EXTREME regimen); nivolumab (second-line)
- Palliative RT: For pain, bleeding, airway obstruction
- Best supportive care: Pain management (WHO analgesic ladder; may need nerve blocks), nutritional support (PEG tube), airway management (tracheostomy), speech therapy, psychological support
- Symptom-specific interventions: Bleeding → palliative RT or embolisation; Dysphagia → stenting or PEG; Fungating wound → regular wound care + metronidazole for odour
Rehabilitation always — swallowing, voice and hearing [3]
| Domain | Intervention |
|---|---|
| Swallowing | Speech and language therapy; swallowing exercises (pre-habilitation before CRT); Modified diet texture; PEG tube if needed |
| Voice | Tracheoesophageal puncture (TEP) with voice prosthesis (gold standard post-laryngectomy); Oesophageal speech; Electrolarynx |
| Hearing | Hearing aids (conductive loss from serous otitis media in NPC); Bone-anchored hearing aids |
| Dental | Obturators for palatal defects; Dental implants post-mandibular reconstruction; Fluoride trays for xerostomia |
| Nutritional | Dietitian input; Enteral feeding if unable to maintain oral intake |
| Psychological | Body image counselling; Depression/anxiety screening; Support groups |
| Physiotherapy | Shoulder rehabilitation post-neck dissection (especially if CN XI sacrificed); Trismus exercises |
- Standard: Clinical examination every 1–3 months in Year 1–2, every 2–4 months in Year 3, every 4–6 months in Years 4–5, then annually
- Imaging: CT/MRI at 3 months post-treatment as new baseline; PET-CT at 12 weeks post-CRT to assess response
- NPC-specific: Post-treatment plasma EBV DNA levels evaluate treatment response and detection of recurrence [2]
- Panendoscopy: Annual for field cancerization surveillance (synchronous/metachronous primaries)
- Thyroid function: Annual TSH if neck irradiated (RT can cause hypothyroidism)
High Yield Summary
Management framework: Early stage (I–II) = single modality; Late stage (III–IV) = combined modality.
Exceptions: Oral cavity → surgery first even in early stage; NPC → chemo-irradiation even in advanced stage.
3 R's of H&N surgery: Resection (oncologically clear margins) → Reconstruction (form and function) → Rehabilitation (swallowing, voice, hearing).
Neck dissection: Selective (elective, levels at risk) vs Modified radical (Levels I–V, preserve structures) vs Radical (Levels I–V + IJV + CN XI + SCM, rarely done).
Larynx management: Early = RT (better voice) or larynx-preserving surgery. Advanced = organ preservation with CRT if candidate; total laryngectomy + post-op RT + voice rehab if not.
NPC management: Stage I = RT only; Stage II–IVB = concurrent CRT ± adjuvant chemo. Surgery NOT first-line (radiosensitive + inaccessible).
Post-op CRT indications: Positive margins and ENE are the two strongest indications (EORTC 22931 / RTOG 9501).
Salivary gland: Surgery first. ACC = complete excision + post-op RT; indolent but late lung metastases. Mucoepidermoid = excision ± RT based on grade. Lymphoepithelial carcinoma = EBV-related, behaves like NPC, good prognosis.
Immunotherapy: Pembrolizumab (anti-PD-1) is now first-line for recurrent/metastatic HNSCC (KEYNOTE-048).
Pre-RT dental assessment mandatory to prevent osteoradionecrosis.
HPV+ oropharyngeal cancer: De-intensification trials ongoing — better prognosis, may not need as aggressive treatment.
Active Recall - Management of Head and Neck Cancer
References
[2] Senior notes: felixlai.md (H&N cancer treatment, CA Oropharynx, NPC, Laryngeal carcinoma, Lip/Tongue/Floor of mouth/Alveolus/Retromolar trigone/Buccal mucosa/Hard palate cancer sections) [3] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (pp. 42, 43, 44, 48) [5] Lecture slides: GC 187. Head and neck cancer problems Function and shape.pdf (pp. 13, 14, 27) [7] Lecture slides: GC 217. Facial nerve palsy and salivary gland diseases.pdf (pp. 67, 69, 74, 76) [8] Image credit: ResearchGate (TORS image)
Complications of Head and Neck Cancer
Complications in H&N cancer arise from three sources: the disease itself (tumour-related), the treatment (surgery, radiotherapy, chemotherapy), and the interaction between these. Understanding complications from first principles requires you to think anatomically — what structures are being damaged, and what functions do those structures serve?
Head and Neck cancer: problems [5]:
- Most frequently exposed region of the body
- Anatomical disruption will affect morphology and physiology
This one slide captures everything. The head and neck is where form meets function. Every complication can be traced back to disruption of structure (morphology) or disruption of what that structure does (physiology).
1. Disease-Related Complications (From the Tumour Itself)
These are complications that arise because the cancer grows, invades, obstructs, or metastasises.
Airway [5]:
- Temporary — swelling
- Permanent — tumour / stricture
Why? Tumours of the larynx (especially supraglottic and subglottic), hypopharynx, tongue base, and oropharynx can grow large enough to physically obstruct the airway lumen. Remember that stridor indicates > 50% luminal obstruction — by the time you hear stridor, the situation is already critical.
Mechanism:
- Direct tumour bulk — endoluminal mass narrows the airway
- Vocal cord fixation — tumour invading the recurrent laryngeal nerve or cricoarytenoid joint causes cord immobility. If bilateral, the cords fix in the adducted (closed) position → complete obstruction
- Peritumoral oedema and bleeding — can cause acute-on-chronic obstruction
Management: ALWAYS protect the airway for all H&N cancer [2]. Emergency tracheostomy or intubation if acute obstruction; elective tracheostomy before definitive treatment if airway is borderline.
Swallowing [5]:
- Voluntary phase usually affected by tumours in the head and neck region
- Usually immediately after glossectomy or pharyngectomy
- Sometimes delayed presentation after radiotherapy
Why? The voluntary (oral preparatory and oral propulsive) phase of swallowing requires intact tongue mobility, buccal function, palatal closure, and pharyngeal constriction. Tumours of the tongue, floor of mouth, oropharynx, and hypopharynx directly impair these mechanics.
Consequences:
- Progressive dysphagia → reduced oral intake → weight loss → cancer cachexia
- Dehydration
- Aspiration pneumonia (when swallowing mechanism fails to protect the airway)
When the tumour disrupts the normal swallow mechanism (laryngeal elevation, epiglottic deflection, vocal cord closure), food and saliva enter the trachea → aspiration → pneumonia. This is particularly common in:
- Supraglottic tumours (epiglottis/aryepiglottic fold involved)
- Tongue base tumours (loss of tongue base retraction during swallowing)
- Post-treatment (after glossectomy, partial laryngectomy, or CRT)
Tumours can erode into blood vessels → bleeding. The severity depends on which vessel is involved:
- Minor: Mucosal bleeding from tumour surface (epistaxis in NPC, blood-stained saliva in oral cancers)
- Major/Life-threatening: Carotid artery blow-out — tumour erodes into the carotid artery (or post-treatment with wound breakdown over the carotid). This is a catastrophic emergency with massive haemorrhage. Risk factors: previous surgery + RT, wound infection/fistula, recurrent tumour encasing the carotid.
NPC can invade the skull base and enter the cavernous sinus or jugular foramen → progressive cranial neuropathies (CN III–VI, IX–XII, sympathetic chain). These are complications of the disease itself and indicate advanced local extension.
- NPC: Bone (75%), liver, lung, distant LN [2]
- Other H&N cancers: Lung > bone > liver
- Complications of distant metastases: pathological fractures (bone), hepatic failure (liver), respiratory failure (lung)
This is a unique "complication" of H&N cancer — not from the original tumour spreading, but from the field cancerization effect [2]:
- Synchronous tumour = Second primary tumour detected within 6 months
- Metachronous tumour = Second primary tumour detected > 6 months
- Oral cavity/oropharynx tumours → second primary in upper oesophagus
- Laryngeal tumours → second primary in lung
This is why lifelong surveillance and panendoscopy are necessary.
2. Surgery-Related Complications
| Complication | Mechanism | Notes |
|---|---|---|
| Bleeding / Haematoma | Reactionary (< 24h) or secondary (> 24h, infection-related) bleeding from surgical bed | Haematoma in the neck is potentially fatal — can compress venous return → laryngeal oedema → airway obstruction → asphyxiation [8]. First action: remove all skin sutures at the bedside to decompress, then resuscitate and return to theatre for haemostasis [8] |
| Wound infection | Oral cavity surgery involves a "clean-contaminated" field (the mouth is colonised) → higher infection risk than clean surgery | Prophylactic antibiotics covering oral flora (e.g., co-amoxiclav) |
| Flap failure | Microvascular free flap — thrombosis of arterial inflow or venous outflow → flap necrosis | Occurs in ~3–5% of free flaps. Requires close monitoring (colour, temperature, capillary refill) in the first 48–72 hours. Early return to theatre for re-exploration if suspected |
2.2 Specific Complications by Surgical Procedure
This is one of the most important sections for exams because total laryngectomy has profound and permanent consequences.
| Complication | Pathophysiology |
|---|---|
| Loss of ability to speak [2] | The larynx is removed → the vibrating vocal cords are gone. Voice rehabilitation is essential (tracheoesophageal puncture with voice prosthesis, oesophageal speech, or electrolarynx) |
| Loss of coughing effort [2] | The glottis normally closes to build subglottic pressure for an effective cough (Valsalva mechanism). Without a larynx, the patient cannot generate this pressure → weak cough → retained secretions. The permanent tracheostomy bypasses the upper airway completely |
| Swallowing dysfunction [2] — Stricture of neopharynx or anastomosis / Regurgitation | The pharynx is reconstructed (neopharynx) and the anastomosis site can stenose from scarring → progressive dysphagia. Managed with serial dilatation |
| Endocrine dysfunction [2] — Hypothyroidism / Hypoparathyroidism | Part or all of the thyroid gland and associated parathyroid glands may be removed depending on extent of disease [2]. Loss of thyroid → hypothyroidism (requires lifelong thyroxine). Loss of parathyroids → hypocalcaemia (perioral numbness, carpopedal spasm, Chvostek's sign, Trousseau's sign; severe cases → laryngospasm [8]) |
| Permanent tracheostomy | Upper aerodigestive tract and digestive tract are permanently disconnected [2] → patient breathes through a stoma. Stigmatisation with the presence of permanent tracheostomy [2] — significant psychological and social impact |
| Pharyngocutaneous fistula | Breakdown of the pharyngeal closure → saliva leaks through the neck wound onto the skin. One of the most common early complications. Risk factors: previous RT, malnutrition, positive margins |
Complications [2]:
- Laryngocutaneous fistula — communication between laryngeal lumen and skin
- Aspiration pneumonia — partial laryngectomy preserves some laryngeal framework but may compromise the sphincteric protective function → chronic aspiration
- Swallowing difficulties
- Bleeding and infection
| Complication | Mechanism |
|---|---|
| Spinal accessory nerve (CN XI) injury | Intentional sacrifice in radical neck dissection, or inadvertent injury during modified radical/selective dissection → trapezius denervation → shoulder drop, chronic pain, inability to abduct shoulder above 90° (shoulder syndrome). This is the most significant morbidity of neck dissection |
| Internal jugular vein (IJV) ligation | If bilateral radical neck dissection removes both IJVs → severe facial oedema, raised intracranial pressure. This is why bilateral radical neck dissection is rarely performed simultaneously |
| Marginal mandibular nerve injury | Branch of CN VII; runs superficially near the submandibular gland. Damage → drooping of ipsilateral lower lip. This is relevant during Level I dissection |
| Hypoglossal nerve (CN XII) injury | Runs deep to the posterior belly of digastric. Damage → ipsilateral tongue deviation and atrophy → impaired speech and swallowing |
| Phrenic nerve injury | C3–C5; runs on the anterior surface of scalenus anterior. Rare but serious → ipsilateral hemidiaphragm paralysis → respiratory compromise |
| Thoracic duct injury | Left Level IV/V dissection can injure the thoracic duct → chylous fistula (milky drainage from neck wound). Managed conservatively (pressure dressing, medium-chain triglyceride diet) or surgically (ligation) if persistent |
| Carotid artery injury | Rare during surgery; more common as a late complication when post-operative wound breakdown exposes the carotid → "carotid blow-out" |
| Lymphoedema | Disruption of cervical lymphatic drainage → facial/submental oedema |
Complications [2]:
- Facial nerve injury — the facial nerve (CN VII) runs through the parotid gland. Even in benign tumour surgery, the nerve must be carefully dissected. Temporary neuropraxia is common (10–20%); permanent injury is rarer but more likely with malignant tumours requiring nerve sacrifice
- Frey syndrome [2]:
- Also known as auriculotemporal syndrome or gustatory sweating
- Characterised by sweating and flushing of facial skin over parotid bed and neck during mastication
- Result of aberrant regeneration of cut parasympathetic fibres between the otic ganglion and salivary tissues which leads to innervation of sweat glands and subcutaneous vessels [2]
Why does Frey syndrome happen? During parotidectomy, the auriculotemporal nerve (a branch of CN V3 carrying parasympathetic fibres to the parotid from the otic ganglion) is divided. During wound healing, regenerating parasympathetic nerve fibres that were destined for the parotid gland instead grow into the cut ends of sympathetic nerves supplying sweat glands in the overlying skin. So when the patient eats (stimulus that should trigger salivation), they instead sweat and flush over the parotid region — the wrong effector organ gets activated. Treatment options include antiperspirants (aluminium chloride), botulinum toxin injection (blocks cholinergic transmission at the sweat gland), or interposition of tissue (e.g., sternocleidomastoid muscle flap) between the parotid bed and the skin during surgery.
| Complication | Mechanism |
|---|---|
| Impaired speech/articulation | Resection of large tumours of tongue that lead to decreased lingual contact with palate, lip, and teeth will result in impaired articulation [2]. The tongue is the primary articulator for consonant sounds (t, d, n, l, s, z) |
| Impaired swallowing | Loss of tongue bulk and mobility → cannot form bolus, cannot propel it posteriorly. Reconstruction using soft pliable fasciocutaneous free flaps can provide intraoral bulk and preservation of tongue mobility [2] |
| Trismus | Fibrosis of pterygoid muscles or temporomandibular joint from surgery ± RT |
| Oro-antral / Oro-nasal fistula | After maxillectomy or palatal resection → communication between oral and nasal/sinus cavities → nasal regurgitation. Through-and-through defects of palate require dental prosthesis [2] |
| Mandibular discontinuity | After segmental mandibulectomy → loss of mandibular arch continuity → cosmetic deformity, malocclusion, difficulty chewing. Reconstructed with fibula free flap |
3. Radiotherapy-Related Complications
Radiotherapy damages both tumour cells and surrounding normal tissue. The complications depend on which normal structures are in the radiation field. Think of RT complications in acute (during/shortly after RT) and late (months to years after RT) categories.
| Complication | Pathophysiology |
|---|---|
| Radiation dermatitis [2] | Direct radiation damage to skin → erythema → dry desquamation → moist desquamation (blistering, peeling). Like a severe sunburn. The skin in the radiation field becomes red, tender, and eventually may ulcerate |
| Mucositis | Radiation destroys rapidly dividing mucosal epithelial cells → painful ulceration of the oropharyngeal mucosa. This is the most debilitating acute side effect — patients cannot eat, drink, or swallow their own saliva. Often requires PEG tube placement for nutrition. Mouth and throat sores [2] |
| Odynophagia [2] | Direct consequence of mucositis → severe pain on swallowing |
| Dysphagia [2] | Mucositis + oedema → difficulty swallowing |
| Hoarseness [2] | Laryngeal oedema from RT to the larynx |
| Taste alteration (dysgeusia) | Radiation damages taste buds on the tongue → altered or absent taste. Usually begins within 1–2 weeks and can last months |
| Nausea | If posterior fossa/brainstem is in the field (NPC) |
| Complication | Pathophysiology |
|---|---|
| Xerostomia (dry mouth) | Radiation damages serous acini of salivary glands (parotid is most sensitive because it is predominantly serous). Salivary flow decreases → dry mouth → difficulty eating, speaking, sleeping; rapid dental caries. Modern IMRT techniques aim to spare the contralateral parotid to preserve some salivary function. This is the most common chronic complaint after H&N RT |
| Osteoradionecrosis (ORN) of the mandible | Radiation causes endarteritis obliterans (obliteration of small blood vessels in bone) → the mandible becomes hypovascular, hypocellular, and hypoxic (the "3H's" of ORN). If the irradiated bone is traumatised (e.g., tooth extraction), it cannot mount a healing response → chronic, painful, non-healing bone exposure. This is why pre-RT dental assessment and extraction of carious teeth is mandatory |
| Laryngeal oedema [2] | Chronic oedema of the laryngeal mucosa from radiation-induced lymphatic damage → voice change, airway compromise |
| Laryngeal or pharyngeal stenosis [2] | Late fibrosis of irradiated tissue → progressive narrowing of the airway or pharynx → stridor, dysphagia |
| Radiation fibrosis / Trismus | Fibrosis of muscles of mastication (especially medial and lateral pterygoids) → progressive inability to open the mouth. Can be very debilitating; managed with jaw-stretching exercises (TheraBite) |
| Hypothyroidism | If the thyroid gland is in the RT field (common in laryngeal, hypopharyngeal RT, and NPC bilateral neck irradiation) → progressive thyroid gland destruction → hypothyroidism. Annual TSH monitoring is mandatory for all H&N RT patients |
| Dental caries (radiation caries) | Consequence of xerostomia — reduced salivary flow removes the protective buffering/antimicrobial effect of saliva → rampant dental decay, especially at the cementoenamel junction. Fluoride trays and meticulous dental hygiene are essential |
| Carotid artery stenosis | Radiation accelerates atherosclerosis in the carotid arteries → increased stroke risk years after RT. Doppler surveillance may be indicated |
| Radiation-induced second malignancy | Radiation is itself a carcinogen. Years after RT, a new cancer can develop within the irradiated field (e.g., sarcoma, new SCC). Rare but well-documented; latency typically > 5 years |
| Hearing loss (sensorineural) | If cochlea is in the field (NPC RT) → radiation cochleitis → high-frequency sensorineural hearing loss. Compounded by cisplatin ototoxicity |
Osteoradionecrosis — Prevention is Key
ORN is far easier to prevent than to treat. All patients must have a dental assessment before starting RT. Carious teeth in the radiation field must be extracted and allowed to heal (minimum 2–3 weeks) before RT begins. After RT, any required extractions should be done with extreme caution — hyperbaric oxygen (HBO) therapy may be used prophylactically in high-risk patients.
Cisplatin side effects [2]:
- Nephrotoxicity — cisplatin is directly toxic to renal tubular cells. Aggressive IV hydration before and after each cycle is mandatory. Monitor creatinine and GFR. If GFR < 50, switch to carboplatin
- Ototoxicity — damage to outer hair cells of the cochlea → bilateral high-frequency sensorineural hearing loss (irreversible). Particularly problematic when combined with RT to the cochlea (NPC). Audiometric monitoring required
- Nausea and vomiting (76–100%) [2] — cisplatin is one of the most emetogenic chemotherapy agents. Requires triple antiemetic prophylaxis (5-HT3 antagonist + NK1 antagonist + dexamethasone)
- Peripheral neuropathy — dose-dependent, cumulative, "stocking-glove" distribution
- Myelosuppression (less marked than with other agents but still significant)
Fluorouracil (5-FU) side effects [2]:
- Myelosuppression (Pancytopenia) — suppresses rapidly dividing bone marrow progenitors → neutropenia (infection risk), anaemia (fatigue), thrombocytopenia (bleeding)
- Alopecia — hair follicle cells are rapidly dividing and are collateral damage
- Stomatitis — 5-FU damages oral mucosal cells → painful mouth ulcers (compounding RT mucositis)
- Diarrhoea — GI mucosal cells are rapidly dividing → damage → secretory diarrhoea. Can be severe and life-threatening
Immune checkpoint inhibitors (pembrolizumab, nivolumab) "release the brakes" on the immune system. The downside is that the unleashed immune system can attack normal tissues → immune-related adverse events (irAEs):
| irAE | Mechanism |
|---|---|
| Pneumonitis | T-cell-mediated lung inflammation → cough, dyspnoea. Can be fatal. Requires steroids |
| Hepatitis | Immune-mediated hepatocyte damage → elevated transaminases |
| Colitis | Immune-mediated GI mucosal inflammation → diarrhoea, abdominal pain |
| Thyroiditis / Hypothyroidism | Immune destruction of thyroid → transient thyrotoxicosis → hypothyroidism |
| Dermatitis | Pruritic maculopapular rash |
| Hypophysitis | Inflammation of pituitary → pan-hypopituitarism (ACTH deficiency is most dangerous → adrenal crisis) |
Management: mild irAEs → hold immunotherapy; moderate to severe → systemic corticosteroids; severe/refractory → immunosuppressants (infliximab, mycophenolate).
These are often underappreciated but profoundly impact quality of life.
Rehabilitation always — swallowing, voice and hearing [3]
| Domain | Complication | Pathophysiology |
|---|---|---|
| Speech | Dysarthria, aphonia | Loss of tongue bulk (glossectomy), loss of palatal closure (maxillectomy), loss of vocal cords (laryngectomy) |
| Swallowing [5] | Voluntary phase usually affected by tumours in the head and neck region. Usually immediately after glossectomy or pharyngectomy. Sometimes delayed presentation after radiotherapy | Loss of tongue mobility, pharyngeal stenosis, reduced laryngeal elevation, fibrosis, xerostomia |
| Breathing | Permanent tracheostomy dependency | After total laryngectomy — upper airway and digestive tract permanently disconnected |
| Cosmetic disfigurement | Facial asymmetry, mandibular defects, neck scarring | Resection of visible structures → psychosocial distress, social isolation |
| Depression and anxiety | Reactive to diagnosis, disfigurement, functional loss, social isolation | Very common; screening essential; refer to clinical psychology |
| Body image disturbance | Loss of facial integrity, permanent stoma, scarring | Especially after extensive resections; compounded by weight loss |
| Social isolation | Difficulty speaking → social withdrawal; permanent tracheostomy → stigmatisation [2]; eating difficulties → cannot socialise over meals |
| Source of Complication | Acute | Late/Chronic |
|---|---|---|
| Disease | Airway obstruction, haemorrhage, aspiration pneumonia, dysphagia, cranial nerve palsies | Distant metastasis, second primary tumours (field cancerization) |
| Surgery | Haematoma (airway emergency), flap failure, wound infection, nerve injury (CN VII, XI, XII, RLN, marginal mandibular), chylous fistula, pharyngocutaneous fistula | Frey syndrome, shoulder syndrome (CN XI), trismus, speech/swallowing impairment, endocrine dysfunction (hypothyroidism, hypoparathyroidism), permanent tracheostomy, stricture |
| Radiotherapy | Mucositis, dermatitis, odynophagia, dysgeusia, hoarseness, laryngeal oedema | Xerostomia, ORN of mandible, fibrosis/trismus, laryngeal/pharyngeal stenosis, dental caries, hypothyroidism, carotid stenosis, radiation-induced second malignancy, SNHL |
| Chemotherapy | N&V, mucositis/stomatitis, myelosuppression, nephrotoxicity (cisplatin), diarrhoea (5-FU) | Ototoxicity (cisplatin), peripheral neuropathy, alopecia |
| Immunotherapy | Rash, colitis, hepatitis, pneumonitis | Thyroiditis/hypothyroidism, hypophysitis |
| Functional/Psychosocial | — | Depression, body image disturbance, social isolation, dysphagia requiring long-term PEG, permanent stoma |
High Yield Summary
Disease complications: Airway obstruction (always protect the airway!), aspiration pneumonia, haemorrhage (carotid blow-out is catastrophic), cranial nerve palsies (NPC), distant metastasis, second primary tumours (field cancerization).
Surgical complications:
- Total laryngectomy: loss of speech, loss of cough effort, swallowing dysfunction (neopharyngeal stricture), endocrine dysfunction (hypothyroidism, hypoparathyroidism), permanent tracheostomy stigmatisation, pharyngocutaneous fistula.
- Partial laryngectomy: aspiration pneumonia, laryngocutaneous fistula, swallowing difficulty.
- Neck dissection: CN XI injury (shoulder drop), chylous fistula (thoracic duct injury), marginal mandibular nerve injury.
- Parotid surgery: CN VII injury, Frey syndrome (aberrant parasympathetic regeneration → gustatory sweating).
- Post-operative haematoma: neck haematoma → venous compression → laryngeal oedema → airway obstruction → asphyxiation. First action = remove sutures at bedside.
RT complications (acute): Mucositis (most debilitating), radiation dermatitis, odynophagia, dysphagia, hoarseness.
RT complications (late): Xerostomia (most common chronic complaint), osteoradionecrosis (prevented by pre-RT dental assessment), fibrosis/trismus, hypothyroidism (annual TSH), laryngeal/pharyngeal stenosis, radiation-induced second malignancy.
Chemotherapy: Cisplatin = nephrotoxicity + ototoxicity + severe nausea. 5-FU = myelosuppression + stomatitis + diarrhoea.
Functional: Speech, swallowing, breathing, cosmesis, and psychosocial impact are unique to H&N cancer and require dedicated rehabilitation.
Active Recall - Complications of Head and Neck Cancer
References
[2] Senior notes: felixlai.md (H&N cancer overview, NPC, Laryngeal carcinoma — treatment/complications, Parotid surgery complications, Thyroidectomy complications, Tongue/Floor of mouth/Palate cancer sections) [3] Lecture slides: GC 219. Infections and tumours in pharynx and oral cavity.pdf (p. 44) [5] Lecture slides: GC 187. Head and neck cancer problems Function and shape.pdf (pp. 4, 7, 9, 13, 14, 27) [8] Senior notes: maxim.md (Thyroidectomy complications — reactionary haemorrhage, RLN injury, parathyroid injury)
High Yield Summary
Definition: H&N cancers are predominantly SCC arising from the mucosal surfaces of the UADT — oral cavity, pharynx (naso-, oro-, hypo-), larynx, nasal cavity/paranasal sinuses, and salivary glands.
Epidemiology: Male predominance, age > 60 (except HPV-related oropharyngeal cancer in younger males). NPC is endemic in Southern China/Hong Kong.
4 Major Risk Factors: HPV (oropharynx) + EBV (NPC) + Smoking + Alcohol. Mnemonic: 5 S's — Smoking, Spirits, Sharp teeth, Sex (male/oral), Spicy food.
Field Cancerization: Diffuse carcinogen exposure → synchronous/metachronous tumours. Always do panendoscopy (direct laryngoscopy + bronchoscopy + OGD).
HPV Mechanism: E6 degrades p53, E7 inactivates Rb → better prognosis, de-intensification possible.
EBV Mechanism: LMP1 activates NF-κB → NPC. Plasma EBV DNA used for screening/monitoring.
Premalignant Lesions: Erythroplakia > Speckled leukoplakia > Leukoplakia in malignant potential.
Key Presentations by Site:
- Lip: Non-healing ulcer on vermilion border (UV exposure)
- Oral tongue: Lateral/ventral ulcer; lingual nerve (paraesthesia), CN XII (deviation)
- NPC: Unilateral serous otitis media, epistaxis, cranial nerve palsies, bilateral neck nodes
- Oropharynx: Sore throat, referred otalgia, dysphagia, muffled voice, 50% cervical LN
- Hypopharynx: Globus → dysphagia, otalgia, hoarseness, loss of laryngeal crepitus
- Glottic larynx: Early hoarseness (most common laryngeal cancer, best prognosis)
- Supraglottic/Subglottic: Late presentation, worse prognosis
Lymphatic Drainage: Glottic = sparse (low nodal risk); Supraglottic = rich (30–50% nodal); Tongue = skip metastasis to Level III–IV.
Always protect the airway.
High Yield Summary
The Big Three DDx Categories: Infective (acute, febrile) vs Neoplastic (chronic, afebrile) vs Congenital/Developmental.
90% of H&N malignancies are SCC (excluding nasopharynx and thyroid).
Oral cavity/oropharynx histological DDx: SCC (epithelial, ulcerative) vs Lymphoma/minor salivary gland tumour (submucosal, smooth).
Key mimics of cancer: Lymphoma (tonsil/tongue base), syphilitic chancre (oral ulcer), branchial cleft cyst (cystic neck mass — may be HPV+ metastasis).
Red flags for malignancy referral: Persistent > 2–4 weeks, irregular, indurated, > 2cm, associated cervical LN. Hoarseness/dysphagia/otalgia with a neck mass suggests metastatic UADT cancer.
FNA limitations: Cannot provide tissue architecture → cannot subtype lymphoma. Need core/excisional biopsy.
Facial nerve palsy with parotid mass = malignant until proven otherwise.
15–20% occult nodal metastasis in oral cavity SCC → elective neck dissection.
Field cancerization → 10% risk synchronous/metachronous tumours → always panendoscopy.
Cystic Level II node in young adult: think HPV+ oropharyngeal SCC, not just branchial cleft cyst.
High Yield Summary
Diagnosis is histological — always need tissue (biopsy/FNA). Cannot diagnose on imaging alone.
Workup sequence: History + PE → Endoscopy + Biopsy → USG + FNAC → CT/MRI → PET-CT if necessary → TNM staging → MDT.
FNA: First-line for neck mass. Can do cytology + HPV/EBV PCR. Cannot subtype lymphoma (no architecture).
Panendoscopy (laryngoscopy + bronchoscopy + OGD): Mandatory for all UADT cancers — 10% synchronous/metachronous tumour risk from field cancerization.
CT vs MRI: CT for bone and speed; MRI for soft tissue, perineural spread, skull base. MRI is imaging of choice for oral cavity/oropharynx; CT crucial for larynx (cartilage invasion).
PET-CT: Superior to CT/MRI for nodal metastasis, distant metastasis, and second primaries. Used for unknown primary and post-treatment surveillance.
NPC workup: Nasopharyngoscopy-guided biopsy (NOT nodal biopsy/dissection); Plasma EBV DNA for monitoring; EBV serology for screening (low specificity).
TNM staging: T = site-specific; N = uniform (except NPC); M = uniform. ENE upstages N category. HPV+ oropharyngeal cancer has separate staging (AJCC 8th ed).
Unknown primary: FNA → p16/EBV testing → PET-CT → Panendoscopy + directed biopsies + bilateral tonsillectomy.
Sonographic features of malignant LN: Size > 1cm short-axis, rounded, loss of fatty hilum, central necrosis, heterogeneous enhancement.
High Yield Summary
Management framework: Early stage (I–II) = single modality; Late stage (III–IV) = combined modality.
Exceptions: Oral cavity → surgery first even in early stage; NPC → chemo-irradiation even in advanced stage.
3 R's of H&N surgery: Resection (oncologically clear margins) → Reconstruction (form and function) → Rehabilitation (swallowing, voice, hearing).
Neck dissection: Selective (elective, levels at risk) vs Modified radical (Levels I–V, preserve structures) vs Radical (Levels I–V + IJV + CN XI + SCM, rarely done).
Larynx management: Early = RT (better voice) or larynx-preserving surgery. Advanced = organ preservation with CRT if candidate; total laryngectomy + post-op RT + voice rehab if not.
NPC management: Stage I = RT only; Stage II–IVB = concurrent CRT ± adjuvant chemo. Surgery NOT first-line (radiosensitive + inaccessible).
Post-op CRT indications: Positive margins and ENE are the two strongest indications (EORTC 22931 / RTOG 9501).
Salivary gland: Surgery first. ACC = complete excision + post-op RT; indolent but late lung metastases. Mucoepidermoid = excision ± RT based on grade. Lymphoepithelial carcinoma = EBV-related, behaves like NPC, good prognosis.
Immunotherapy: Pembrolizumab (anti-PD-1) is now first-line for recurrent/metastatic HNSCC (KEYNOTE-048).
Pre-RT dental assessment mandatory to prevent osteoradionecrosis.
HPV+ oropharyngeal cancer: De-intensification trials ongoing — better prognosis, may not need as aggressive treatment.
High Yield Summary
Disease complications: Airway obstruction (always protect the airway!), aspiration pneumonia, haemorrhage (carotid blow-out is catastrophic), cranial nerve palsies (NPC), distant metastasis, second primary tumours (field cancerization).
Surgical complications:
- Total laryngectomy: loss of speech, loss of cough effort, swallowing dysfunction (neopharyngeal stricture), endocrine dysfunction (hypothyroidism, hypoparathyroidism), permanent tracheostomy stigmatisation, pharyngocutaneous fistula.
- Partial laryngectomy: aspiration pneumonia, laryngocutaneous fistula, swallowing difficulty.
- Neck dissection: CN XI injury (shoulder drop), chylous fistula (thoracic duct injury), marginal mandibular nerve injury.
- Parotid surgery: CN VII injury, Frey syndrome (aberrant parasympathetic regeneration → gustatory sweating).
- Post-operative haematoma: neck haematoma → venous compression → laryngeal oedema → airway obstruction → asphyxiation. First action = remove sutures at bedside.
RT complications (acute): Mucositis (most debilitating), radiation dermatitis, odynophagia, dysphagia, hoarseness.
RT complications (late): Xerostomia (most common chronic complaint), osteoradionecrosis (prevented by pre-RT dental assessment), fibrosis/trismus, hypothyroidism (annual TSH), laryngeal/pharyngeal stenosis, radiation-induced second malignancy.
Chemotherapy: Cisplatin = nephrotoxicity + ototoxicity + severe nausea. 5-FU = myelosuppression + stomatitis + diarrhoea.
Functional: Speech, swallowing, breathing, cosmesis, and psychosocial impact are unique to H&N cancer and require dedicated rehabilitation.
Facial Nerve Palsy
Facial nerve palsy is dysfunction of cranial nerve VII resulting in partial or complete weakness of the muscles of facial expression on the affected side.
Laryngeal Carcinoma
Laryngeal carcinoma is a malignant neoplasm arising from the epithelial lining of the larynx, most commonly squamous cell carcinoma, strongly associated with smoking and alcohol use, presenting with hoarseness, dysphagia, or stridor.